Mechanical Force at the Immunological Synapse: How CD4 T-Cells Sense and Respond to Their Physical Microenvironment
This review synthesizes current research on the mechanical regulation of CD4 T-cell activation through the immunological synapse (IS).
Mechanical Force at the Immunological Synapse: How CD4 T-Cells Sense and Respond to Their Physical Microenvironment
Abstract
This review synthesizes current research on the mechanical regulation of CD4 T-cell activation through the immunological synapse (IS). We explore foundational biophysical concepts of force generation and sensing, detail cutting-edge methodologies for measuring and manipulating the synaptic mechanical environment, address common experimental challenges, and validate findings through comparative analysis across T-cell subsets and disease models. Aimed at researchers and drug developers, this article provides a comprehensive framework for understanding mechano-immunology's role in adaptive immunity and its therapeutic potential.
The Biomechanical Blueprint: Understanding Force Transmission in the CD4 T-Cell Synapse
This whitepaper provides an in-depth technical guide to the immunological synapse (IS), the specialized supramolecular structure that forms at the contact site between a T-cell and an antigen-presenting cell (APC). The discussion is framed within the context of a broader thesis investigating how the mechanical properties of the extracellular environment influence CD4+ T-cell activation, signaling fidelity, and effector function via IS maturation and stability.
Architecture of the Immunological Synapse
The canonical bullseye model comprises concentric rings: a central supramolecular activation cluster (cSMAC) surrounded by the peripheral (pSMAC) and distal (dSMAC) regions.
Table 1: Architectural Zones of the Immunological Synapse
| Zone | Key Molecular Components | Primary Function | Diameter (µm) |
|---|---|---|---|
| cSMAC | TCR-pMHC, CD28, PKCθ, CD3ζ | Sustained TCR signaling termination; internalization of TCR complexes. | 0.5 - 2 |
| pSMAC | LFA-1-ICAM-1, Talin, F-actin | Adhesion stabilization; mechanical force transduction. | 2 - 6 |
| dSMAC | CD45, CD43, F-actin flow | Exclusion of phosphatases; actin retrograde flow (~0.1 µm/s). | 6 - 12 |
Molecular Orchestration of Synapse Formation
The IS assembly is a highly dynamic, actomyosin-driven process. Critical steps include:
- Initial Adhesion: LFA-1/ICAM-1 mediated weak tethering.
- Antigen Recognition: TCR engagement with peptide-MHC (pMHC).
- Microcluster Formation: TCR and signaling molecules aggregate into microclusters.
- Centripetal Transport: Actin retrograde flow transports microclusters inward to form the cSMAC.
- Maturation: Stable polarization of the microtubule organizing center (MTOC) and secretory apparatus toward the IS.
Diagram 1: IS Assembly and Signaling Cascade
The CD4+ T-cell Mechanical Environment and IS Stability
The mechanical stiffness of the APC surface or artificial substrate is a critical regulator of IS function. Studies using polyacrylamide gels or supported lipid bilayers (SLBs) with tunable stiffness demonstrate that intermediate stiffness (∼10-50 kPa) optimizes CD4+ T-cell activation by promoting sustained LFA-1 clutch engagement and mechanosensing via the Talin-Vinculin axis.
Table 2: Impact of Substrate Stiffness on CD4+ T-cell IS Metrics
| Substrate Stiffness | IS Area (µm²) | TCR Microcluster Lifetime (s) | Mean Calcium Flux Duration (min) | IL-2 Secretion (pg/cell) |
|---|---|---|---|---|
| Soft (1 kPa) | 45 ± 12 | 85 ± 15 | 12 ± 3 | 0.8 ± 0.2 |
| Medium (25 kPa) | 78 ± 18 | 145 ± 22 | 28 ± 5 | 2.5 ± 0.6 |
| Stiff (100 kPa) | 65 ± 15 | 110 ± 18 | 18 ± 4 | 1.2 ± 0.3 |
Experimental Protocols for IS Analysis
Protocol 4.1: High-Resolution Imaging of IS on Supported Lipid Bilayers
Objective: To visualize real-time dynamics of protein organization in the IS.
- SLB Preparation: Fuse small unilamellar vesicles containing ICAM-1 and pMHC monomers onto a clean glass coverslip in a flow chamber.
- T-cell Preparation: Isolate naïve CD4+ T-cells from mouse spleen or human PBMCs. Label surface proteins (e.g., TCR with fluorescent antibody) or transfect with GFP-tagged constructs (e.g., ZAP70-GFP).
- Image Acquisition: Introduce cells into the chamber on a confocal or TIRF microscope maintained at 37°C/5% CO2. Acquire time-lapse images every 5-10 seconds for 20-30 minutes.
- Analysis: Use particle tracking software (e.g., TrackMate in Fiji) to quantify microcluster velocity and lifetime.
Protocol 4.2: Measuring Molecular Forces via FRET-based Tension Sensors
Objective: Quantify piconewton-scale forces across specific IS molecules (e.g., TCR, LFA-1).
- Sensor Incorporation: Use SLBs or functionalized beads presenting pMHC/ICAM-1 conjugated with a tension sensor module (e.g., TSMod), which contains donor and acceptor fluorophores linked by an extensible peptide.
- T-cell Engagement: Allow T-cells to engage the substrate.
- FRET Measurement: Acquire donor and acceptor emission signals using ratiometric imaging. A decrease in FRET efficiency correlates with force-induced extension of the peptide linker.
- Calibration: Convert FRET ratio to force (pN) using a calibrated standard curve.
Diagram 2: FRET-based Tension Sensor Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for IS Research
| Item / Reagent | Function / Application | Example Product / Citation |
|---|---|---|
| Supported Lipid Bilayers (SLBs) | Presents mobile antigens and adhesion molecules in a planar format to mimic APC surface. Enables high-resolution imaging. | Formulated in-house with purified ICAM-1 & pMHC; or commercial vesicles (e.g., Avanti Polar Lipids). |
| DNA-based Tension Gauge Tether (TGT) | Measures integrin forces by presenting ligand on a DNA strand with a defined unzipping force threshold. | Custom synthesized oligos with thiol and biotin modifications for surface conjugation. |
| Photoactivatable pMHC (pa-pMHC) | Allows precise spatial and temporal uncaging of antigen with UV light to initiate synapse formation at a defined site. | Developed in-house via site-specific conjugation of photocleavable groups to pMHC. |
| Inhibitors:- PP2 (Src kinase inhibitor)- Latrunculin A (Actin depolymerizer)- Blebbistatin (Myosin II inhibitor) | Perturb specific signaling or cytoskeletal pathways to dissect their contribution to IS architecture and function. | Available from major suppliers (e.g., Sigma-Aldrich, Tocris). |
| Live-cell dyes:- Fluo-4 AM (Calcium)- Cell Tracker dyes | Report on intracellular signaling dynamics and allow for cell discrimination in co-culture experiments. | Available from Thermo Fisher Scientific. |
| Anti-CD3/CD28 functionalized magnetic beads | Provides a standardized, artificial APC system to study activation and IS formation in the absence of a true APC. | Dynabeads Human T-Activator CD3/CD28 (Thermo Fisher). |
Signaling Pathways Converging at the Immunological Synapse
Diagram 3: Core TCR Proximal Signaling Network at the IS
The immunological synapse is a dynamically organized signaling hub where architectural patterns and molecular forces are inextricably linked to functional outcomes. For CD4+ T-cells, the mechanical context is not merely a passive substrate but an active participant in shaping signal quality, duration, and ultimately, the adaptive immune response. Advanced tools enabling molecular-scale force measurement and high-resolution spatial-temporal analysis continue to refine our understanding, offering new targets for modulating immune function in therapy.
Mechano-immunology investigates how immune cells sense, generate, and respond to mechanical forces, integrating biophysical principles with immunological function. This field provides a critical framework for understanding the dynamics of the immunological synapse (IS), particularly for CD4+ T-cells. The mechanical properties of the IS—including substrate stiffness, ligand mobility, and cortical tension—are not passive backgrounds but active regulators of T-cell receptor (TCR) signaling, cytoskeletal rearrangement, and ultimate effector functions. This guide details the core principles, quantitative data, and experimental methodologies underpinning this paradigm, contextualized within CD4+ T-cell research.
Core Principles of Force Generation and Sensing
T-cell mechanosensing is a cyclical process: 1. Force Generation: The T-cell actin cytoskeleton, powered by non-muscle myosin IIA (NMIIA), generates contractile forces against the antigen-presenting cell (APC) surface. 2. Force Transmission: Forces are transmitted through surface receptors (e.g., TCR, LFA-1) bound to their cognate ligands (pMHC, ICAM-1) on the APC. 3. Mechanosensing: Conformational changes in strained receptor-ligand bonds (e.g., TCR-pMHC) or associated proteins (e.g., talin, kindlin) expose cryptic binding sites, enhancing downstream signaling. 4. Adaptive Response: Signaling reinforces cytoskeletal polarization and modulates force generation, creating a feedback loop that calibrates the immune response.
Quantitative Data in T-Cell Mechanobiology
Key mechanical parameters and their immunological consequences are summarized below.
Table 1: Quantitative Metrics of T-Cell Force and Substrate Properties
| Parameter | Typical Measured Value / Range | Measurement Technique | Functional Implication for CD4+ T-Cells |
|---|---|---|---|
| TCR Triggering Force | ~10-15 picoNewtons (pN) | Biomembrane Force Probe (BFP), Optical Tweezers | Minimum force required for optimal TCR triggering and downstream Ca²⁺ flux. |
| T-Cell Traction Force | 1-100 nN/µm² | Traction Force Microscopy (TFM) on polyacrylamide gels | Correlates with activation strength; sustains synapse stability. |
| Optimal Substrate Stiffness | ~10-100 kPa (for naïve T-cells) | Atomic Force Microscopy (AFM), TFM | Mimics stiffness of lymphoid tissue; promotes spreading, signaling, and proliferation. |
| TCR-pMHC Bond Lifetime (Catch Bond) | Increases from ~0.5 to ~5 sec under 10-15 pN load | BFP, Single-Molecule Force Spectroscopy | Prolonged engagement under force enhances signal amplification. |
| Actin Retrograde Flow Rate | ~0.1-0.2 µm/sec in periphery | Total Internal Reflection Fluorescence (TIRF) Microscopy | Drives TCR-pMHC transport to the central supramolecular activation cluster (cSMAC). |
Experimental Protocols
Protocol 1: Traction Force Microscopy (TFM) for IS Mechanics
Objective: To quantify the magnitude and spatial distribution of forces exerted by a CD4+ T-cell on a deformable substrate during synapse formation.
- Substrate Preparation: Fabricate polyacrylamide (PAA) hydrogels of defined stiffness (e.g., 5, 50 kPa) doped with 0.2 µm fluorescent carboxylated beads. Functionalize the surface with anti-CD3ε and ICAM-1 using a heterobifunctional crosslinker (e.g., Sulfo-SANPAH).
- Cell Preparation: Isolate primary murine or human naïve CD4+ T-cells. Label actin (e.g., LifeAct-GFP) or specific proteins of interest via transduction.
- Imaging: Plate T-cells on the functionalized gel. Acquire time-lapse images using a confocal or TIRF microscope with environmental control (37°C, 5% CO₂). Capture both bead positions (for displacement) and cell morphology channels.
- Force Calculation:
- Reference Image: After experiment, lyse cells with 1% SDS to obtain the bead positions in the relaxed, unstressed substrate.
- Displacement Field: Calculate the bead displacement field between stressed (during cell contact) and reference states using particle image velocimetry (PIV).
- Traction Stress: Invert the displacement field using a Fourier Transform-based method (e.g., constrained correlation algorithm) to compute the 2D traction stress vectors (τ) at the cell-substrate interface.
Protocol 2: Biomembrane Force Probe (BFP) for Single-Molecule Kinetics
Objective: To measure the force-dependent binding kinetics (catch bond) of individual TCR-pMHC interactions.
- Probe Assembly: A red blood cell (RBC) is aspirated onto a micropipette to form a pressurized, stiffened seal. A glass bead coated with recombinant pMHC is attached to the RBC apex via a flexible PEG tether, creating a spring.
- Target Cell Preparation: A CD4+ T-cell is aspirated onto a separate micropipette. Its surface is functionalized with specific antibodies or retains native TCRs.
- Force Measurement: The pMHC bead is brought into contact with the T-cell using precision micromanipulation. Upon adhesion, the T-cell is retracted at constant speed, loading the bond. The RBC deformation (∆x) is tracked via high-speed video microscopy.
- Data Analysis: Force (F) is calculated as F = k * ∆x, where k is the BFP spring constant. Bond lifetime (τ) is plotted against force (F) to generate a "catch bond" curve, where τ increases over a specific force range before falling off ("slip bond" regime).
Visualizations
Diagram Title: T-Cell Mechano-Immunology Feedback Cycle
Diagram Title: Traction Force Microscopy Protocol Steps
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for Mechano-Immunology
| Item / Reagent | Function in Experiment | Example / Notes |
|---|---|---|
| Polyacrylamide (PAA) Hydrogels | Tunable, deformable substrate for Traction Force Microscopy (TFM). | Stiffness controlled by bis-acrylamide ratio; functionalized with ligands. |
| Functionalization Crosslinker (Sulfo-SANPAH) | Covalently links proteins (e.g., anti-CD3, ICAM-1) to PAA gel surface. | UV-activated for stable, oriented ligand presentation. |
| Fluorescent Microspheres (200nm) | Embedded fiducial markers in TFM gels to track substrate deformation. | Carboxylated beads for gel incorporation; red or far-red emission preferred. |
| Biomembrane Force Probe (BFP) Setup | High-resolution system to measure single-molecule forces and kinetics. | Comprises micropipettes, RBC, pMHC-coated bead, high-speed camera. |
| Myosin II Inhibitor (Blebbistatin) | Pharmacologically inhibits non-muscle myosin IIA to dissect its role. | Used to perturb force generation; control with inactive enantiomer. |
| Talin Knockdown/CRISPR Cells | Genetically modified T-cells to study force transmission adaptor function. | Validates talin's critical role in linking LFA-1 to the actin cytoskeleton. |
| LifeAct- or F-tractin- GFP/RFP | Live-cell fluorescent probes to visualize actin dynamics in real time. | Expressed via nucleofection or viral transduction. |
| CD3/CD28 Activator Beads (Magnetic) | Standardized, stimulatory surfaces with controlled rigidity and ligand density. | Useful for comparative studies of biochemical vs. mechano-stimulation. |
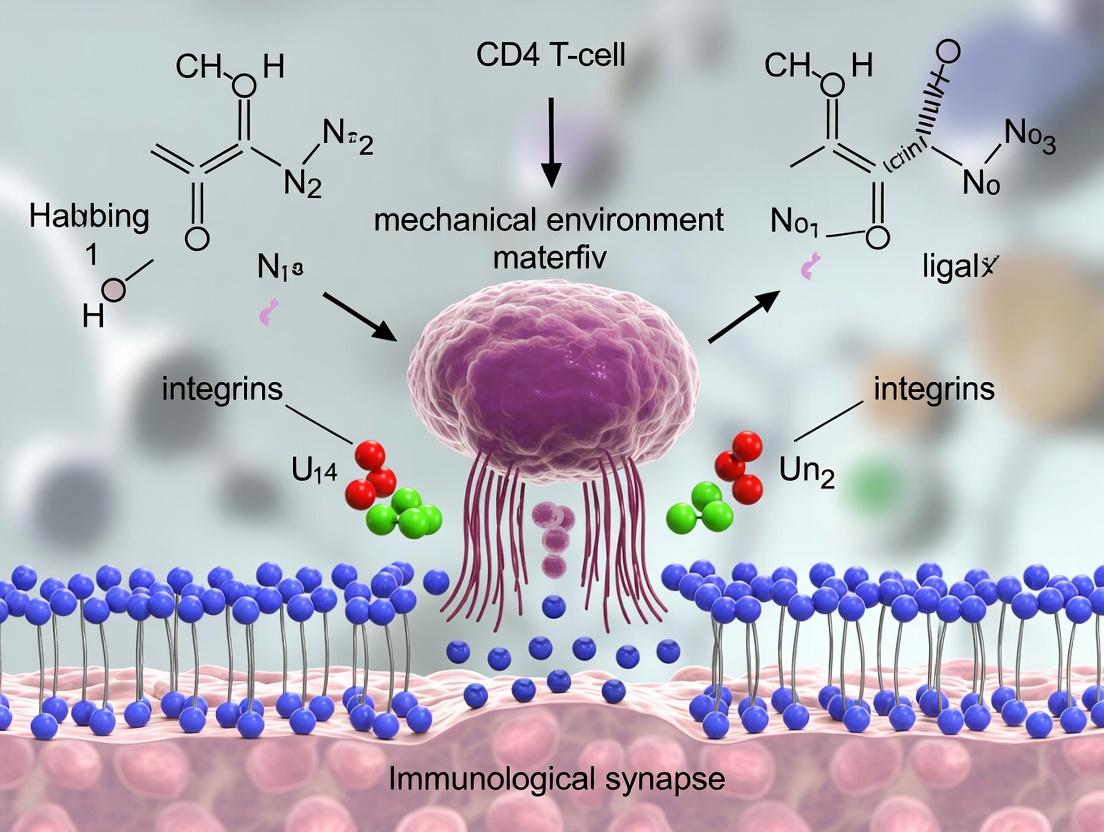
This whitepaper provides an in-depth technical analysis of the core mechanical and biophysical components governing CD4+ T-cell activation within the immunological synapse (IS). Framed within contemporary research on the T-cell mechanical microenvironment, it details the interplay between T-cell receptor (TCR)-peptide-MHC (pMHC) bond mechanics, integrin-mediated adhesion, and actin cytoskeleton dynamics. The integration of these elements determines the biophysical signaling platform, ultimately modulating T-cell functional outcomes. This guide is intended for researchers and drug development professionals exploring mechano-immunology and novel immunotherapeutic targets.
The immunological synapse is a highly structured, dynamic interface between a T-cell and an antigen-presenting cell (APC). Beyond a purely biochemical signaling hub, the IS is a mechanical environment where forces, spatial organization, and rigidity sensing are integral to signal regulation. For CD4+ T-cells, successful activation requires the integration of specific antigen recognition (via TCR-pMHC) with adhesive stability (via integrins, chiefly LFA-1/ICAM-1) and active cytoskeletal remodeling. This document dissects the roles of these three key mechanical players, emphasizing their quantifiable biophysical parameters and interdependent functions within the mechanosensitive framework of CD4+ T-cell activation.
The Core Mechanical Players: Detailed Analysis
TCR-pMHC Bonds: Force-Sensitive Antigen Recognition
The TCR-pMHC interaction is the central antigen-specific signal initiator. Its mechanical properties are critical for ligand discrimination.
- Catch-Bond Behavior: Under physiological tensile force, certain TCR-pMHC bonds exhibit increased lifetime ("catch bonds"), enhancing signal initiation before transitioning to slip-bond behavior at higher forces.
- Mechanical Triggering: Force exerted on the TCR-CD3 complex induces conformational changes and promotes phosphorylation events.
Table 1: Quantitative Biophysical Parameters of Key Interactions
| Interaction | 2D Affinity (KD) | Bond Lifetime (koff) | Force Regime | Characteristic Force (pN) | Key Functional Role |
|---|---|---|---|---|---|
| TCR - agonist pMHC | 1-100 µM | ~0.1 - 10 s (force-modulated) | Catch-bond (~5-15 pN) | ~10-15 pN | Signal triggering, antigen discrimination |
| LFA-1 - ICAM-1 (inactive) | >100 µM | <0.1 s | Slip-bond | N/A | Weak basal adhesion |
| LFA-1 - ICAM-1 (active) | ~0.1-1 µM | ~1-30 s | Catch-bond (~10-40 pN) | ~20-40 pN | Strong adhesion, cytoskeletal coupling |
| Talin - Actin | N/A | N/A | N/A | N/A | Force transmission, integrin activation |
Integrins (LFA-1): Tunable Adhesive Clutches
Lymphocyte function-associated antigen-1 (LFA-1, αLβ2) binding to ICAM-1 on the APC provides the essential adhesive force counterbalance for TCR engagement.
- Inside-Out & Outside-In Signaling: TCR signals rapidly activate LFA-1 via talin and kindlin (inside-out), increasing its affinity. High-affinity LFA-1 then binds ICAM-1, transmitting force back into the cell (outside-in) to stabilize the synapse and reinforce signaling.
- Mechanical Role: Integrins act as a tunable "clutch," linking the retrograde actin flow to the APC surface. This coupling is essential for generating and sustaining the forces needed for TCR scanning and signaling.
The Actin Cytoskeleton: The Active Scaffold
The actin network is the primary force-generating and structural element.
- Retrograde Flow: Centripetal movement of actin from the synapse periphery toward the center, driven by polymerization and myosin II contractility.
- Force Generation & Transmission: The cytoskeleton transmits forces generated internally to TCR and integrin bonds via adaptor proteins (e.g., talin).
- Spatial Organization: Actin clears central TCR microclusters, facilitating signal transduction and creating the canonical bull's-eye pattern of the mature synapse.
Interdependent Signaling and Mechanical Pathways
Diagram Title: TCR Mechanotransduction and Actin Force Coupling Pathway
Key Experimental Methodologies
Two-Dimensional (2D) Micropipette Adhesion Frequency Assay
Purpose: To measure the binding kinetics (2D affinity and kinetics) of TCR-pMHC or LFA-1-ICAM-1 under controlled force. Detailed Protocol:
- Cell Preparation: A red blood cell (RBC) or synthetic bead is coated with purified pMHC or ICAM-1. A CD4+ T-cell (primary or hybridoma) is held by a separate micropipette.
- Contact Control: Using micromanipulators, the APC surrogate is brought into brief (2-10 sec), gentle contact with the T-cell at a predefined area.
- Adhesion Detection: The surfaces are separated. Adhesion is detected by a visible stretching of the RBC membrane or bead displacement.
- Data Acquisition: Repeat for 50-100 cycles. Adhesion frequency (Pa) is calculated.
- Kinetic Analysis: Pa is analyzed as a function of contact time using a probabilistic model to extract the 2D effective on-rate (kon) and off-rate (koff).
- Force Application: Incorporate a force clamp by applying suction pressure to measure koff under defined tensile forces, identifying catch-bond behavior.
Traction Force Microscopy (TFM) on Polyacrylamide Gels
Purpose: To quantify the magnitude and spatial distribution of forces exerted by a T-cell on its substrate via integrins. Detailed Protocol:
- Substrate Fabrication: Prepare flexible polyacrylamide gel (elastic modulus ~10 kPa, mimicking lymphoid tissue) doped with fluorescent carboxylated beads (0.2 µm). Functionalize surface with anti-CD3/CD28 and ICAM-1 via sulfo-SANPAH crosslinking.
- Imaging: Plate CD4+ T-cells onto the gel. Acquire time-lapse images of bead positions using confocal or TIRF microscopy.
- Reference Image: After experiment, lyse the cell (using 1% SDS) to obtain bead positions in the unstressed state.
- Displacement Field Calculation: Use particle image velocimetry (PIV) to compute bead displacement between stressed and reference states.
- Traction Force Calculation: Invert the displacement field using Fourier Transform Traction Cytometry (FTTC) or Bayesian methods to calculate the underlying traction stress vectors (units: Pa).
DNA-Based Tension Gauge Tether (TGT) Assay
Purpose: To probe the specific piconewton (pN) forces exerted by individual receptors (TCR or LFA-1). Detailed Protocol:
- TGT Surface Preparation: Design DNA duplex "springs" with a defined unzipping force threshold (e.g., 12 pN, 19 pN, 54 pN). Conjugate one strand to a surface, the other to a ligand (pMHC or ICAM-1).
- Cell Seeding: Incubate CD4+ T-cells on the TGT surface. If cellular force exceeds the tether's threshold, the duplex unzips, releasing the ligand.
- Detection: Include a fluorescent tag (e.g., Cy3) on the ligand strand. Loss of fluorescence at the cell-substrate interface indicates force transmission through that specific receptor-ligand pair.
- Multiplexing: Use different force thresholds and fluorophores to simultaneously map forces on TCR and LFA-1.
Diagram Title: Integrating Key Mechanobiology Experimental Workflows
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Tools for Mechano-Immunology Research
| Reagent / Tool | Function / Purpose | Example / Specification |
|---|---|---|
| Recombinant pMHC Monomers (Fluorophore-Labeled) | To study TCR-specific binding and kinetics in solution or on surfaces. Essential for tetramer staining and 2D assays. | Human or mouse class II (e.g., HLA-DR, I-Ab) loaded with specific peptide. Biotinylated for surface coupling. |
| ICAM-1 / LFA-1 Blocking Antibodies | To perturb integrin-mediated adhesion and study its specific contribution to synapse mechanics and signaling. | Anti-human CD11a (clone HI111) or anti-CD18 (clone TS1/18). Functional grade, low endotoxin. |
| Flexible Polyacrylamide Gel Kits | To create tunable stiffness substrates for TFM and mechanosensing studies. | Commercial kits (e.g., CytoSoft) or lab-prepared using acrylamide/bis-acrylamide, calibrated with a rheometer. |
| Talin / Vinculin Lifeact Biosensors | To visualize and quantify force-dependent recruitment of cytoskeletal adaptors in live cells. | Lentiviral vectors encoding Lifeact-GFP (F-actin) or Talin-GFP for live-cell TIRF/confocal microscopy. |
| Myosin II Inhibitors (Blebbistatin) | To specifically inhibit non-muscle myosin II contractility, dissecting its role in force generation and actin flow. | (-)-Blebbistatin (active enantiomer) prepared in DMSO; use with dark conditions due to photosensitivity. |
| DNA Oligos for TGTs | To construct tension gauge tethers for measuring single-receptor forces. Requires precise design and conjugation. | HPLC-purified oligonucleotides with thiol, biotin, or DBCO modifications for surface and ligand coupling. |
| Micropipette Puller & Micromanipulator | For performing 2D adhesion assays and single-cell force measurements. | Pipette puller (e.g., Sutter P-97) to create ~1µm tips. Motorized micromanipulators (e.g., Sutter MP-285). |
The mechanical interplay between TCR-pMHC bonds, integrins, and the actin cytoskeleton forms a unified system that CD4+ T-cells exploit to decode antigen quality and environmental stiffness. Disruptions in this mechanical regulation are implicated in autoimmunity, immunodeficiency, and cancer immunotherapy failure. Emerging therapeutic strategies aim to modulate these mechanical players, such as developing allosteric integrin agonists or antagonists, engineering TCR-based therapeutics with optimized mechanosensing properties, and targeting cytoskeletal regulators (e.g., myosin II) to modulate T-cell function in solid tumors. A deep understanding of these core mechanical players is therefore paramount for the next generation of mechano-informed immunotherapies.
The activation of CD4+ T-cells, central to adaptive immunity, occurs at a specialized junction with an antigen-presenting cell (APC) known as the immunological synapse (IS). Traditional biochemical models focus on ligand-receptor interactions. However, emerging research within the broader thesis of the CD4+ T-cell mechanical environment posits that physical cues from the cellular microenvironment are integral to IS formation, signaling fidelity, and functional outcomes. This whitepaper examines how two critical biophysical parameters—substrate stiffness and nanoscale topography—act as co-regulatory "triggers" that modulate mechanotransduction pathways to influence "signal" amplification and T-cell activation.
Substrate Stiffness: A Mechanotransduction Lever
Substrate stiffness mimics the rigidity of target tissues or APCs, providing a mechanical context for T-cell receptor (TCR) engagement.
2.1 Key Quantitative Findings Recent studies quantify the impact of stiffness on T-cell responses using polyacrylamide or polydimethylsiloxane (PDMS) hydrogels of defined Young's modulus.
Table 1: Impact of Substrate Stiffness on CD4+ T-Cell Activation Metrics
| Young's Modulus (kPa) | Proliferation Index | IL-2 Production | Actin Polymerization | NFAT Nuclear Translocation | Primary Reference Model |
|---|---|---|---|---|---|
| ~0.5-1 (Soft) | Low (~1.5x) | Reduced (≤ 30% of max) | Disorganized, slow | Inefficient (~20% cells) | Naive T-cell on soft APC |
| ~10-30 (Intermediate) | High (~4.5x) | Peak (100%) | Robust, centralized | Efficient (~80% cells) | T-cell on dendritic cell |
| ~50-100 (Stiff) | Moderate (~2.8x) | Sustained (~70%) | Over-stabilized, spread | Enhanced but promiscuous | Inflammatory tissue site |
2.2 Signaling Pathways in Stiffness Sensing T-cells sense stiffness via TCR and integrin engagement, triggering actomyosin contractility. Force generation through the TCR-CD3 complex leads to conformational changes in proteins like Talin, exposing binding sites and promoting downstream signaling.
2.3 Experimental Protocol: Measuring T-Cell Activation on Tunable Stiffness Gels
- Materials: Polyacrylamide gel kit, Sulfo-SANPAH crosslinker, recombinant ICAM-1 & anti-CD3ε antibody, murine or human CD4+ T-cells.
- Procedure:
- Prepare gel solutions to achieve 1, 10, and 50 kPa stiffness as validated by rheometry.
- Polymerize gels on activated glass coverslips.
- Functionalize gel surface: Sulfo-SANPAH UV activation, incubate with ICAM-1 (2 μg/mL) and anti-CD3ε (5 μg/mL).
- Isolate naive CD4+ T-cells (e.g., negative selection from mouse spleen).
- Plate cells on gels (50,000 cells/cm²) and incubate (37°C, 5% CO2) for 18-72 hours.
- Assays: Fix for phospho-ZAP70/ERK immunofluorescence; collect supernatant for IL-2 ELISA; use live-cell imaging for actin-GFP dynamics.
Substrate Topography: Nanoscale Geometry as a Signal
Nanotopography (pillars, grooves) influences cytoskeletal patterning, organelle positioning, and receptor clustering at the IS.
3.1 Key Quantitative Findings Studies using electron-beam lithography to create nanoscale arrays reveal how physical constraints shape T-cell responses.
Table 2: Impact of Nanotopography on CD4+ T-Cell Activation Metrics
| Topography Type | Feature Size | TCR Cluster Organization | Microcluster Motility | Mitochondrial Repositioning | Signal Amplification |
|---|---|---|---|---|---|
| Flat 2D Control | N/A | Peripheral, dispersed | Fast (~0.2 μm/s) | Delayed (>30 min) | Baseline |
| Nanoscale Pillars | 100-200 nm diam | Confined to pillar tips | Restricted (~0.05 μm/s) | Enhanced kinetics | ~1.8x pERK vs. control |
| Aligned Nanogrooves | 150 nm width | Linear, aligned | Directed along grooves | Efficient | ~1.5x NFAT vs. control |
| Micron-scale Disordered | 1-2 μm features | Large, irregular aggregates | Arrested | Inconsistent | Suppressed |
3.2 Signaling Pathways in Topography Sensing Nanopatterns direct cytoskeletal forces, segregating inhibitory receptors (e.g., CD45) from TCR microclusters based on spatial exclusion, thereby modulating signal initiation.
3.3 Experimental Protocol: Fabricating and Testing Nanotopographic Substrates
- Materials: Silicon wafer, PMMA or HSQ resist for e-beam lithography, PDMS (Sylgard 184), protein A/G, stimulatory antibodies.
- Procedure:
- Fabrication: Design nanopillar/groove array (e.g., 150 nm diameter, 300 nm pitch). Use e-beam lithography on silicon wafer to create master mold.
- Replication: Cast PDMS (10:1 base:curing agent) onto mold, cure, and peel off.
- Functionalization: Oxygen plasma treat PDMS replica. Incubate with protein A/G (10 μg/mL), then anti-CD3/anti-CD28 antibodies.
- Cell Assay: Seed fluorescently labeled (e.g., CellTracker) CD4+ T-cells. Allow to settle and activate (37°C).
- Imaging: Use high-resolution TIRF or confocal microscopy at 5-20 min intervals to track TCR-GFP or actin-mCherry dynamics. Perform fixed-cell imaging for phospho-specific stains.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Mechano-Immunology Studies
| Reagent / Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Polyacrylamide Gel Kits | BioVision, Cytoskeleton | Provides tunable stiffness substrates for 2D cell culture. |
| PDMS (Sylgard 184) | Dow Corning, Ellsworth | Elastomer for fabricating micro- and nano-topographic replicas; tunable stiffness. |
| Recombinant ICAM-1 | R&D Systems, Sino Biological | Key adhesion ligand for LFA-1 integrin, co-immobilized with TCR stimuli. |
| Anti-CD3ε (Functional Grade) | BioLegend, Tonbo Biosciences | Immobilized antibody for specific TCR cross-linking and activation. |
| Actin Live-Cell Dyes | Thermo Fisher (SiR-actin), Cytoskeleton | Real-time visualization of cytoskeletal dynamics in response to biophysical cues. |
| Phospho-Specific Antibodies | Cell Signaling Technology | Detection of key signaling node activation (e.g., pZAP70, pERK, pLAT). |
| NFAT Translocation Reporter | Addgene (plasmid), Invitrogen (cell line) | Genetically encoded reporter for quantifying downstream transcriptional activation. |
| E-Beam Resist (HSQ/PMMA) | MicroChem, Kayaku | High-resolution resist for creating nanotopography master molds via electron-beam lithography. |
The integration of substrate stiffness and topography presents a mechanical code that CD4+ T-cells decipher at the immunological synapse. Stiffness governs the magnitude of actomyosin-driven force, while topography dictates the spatial organization of signaling components. Future drug development, particularly for immuno-oncology and autoimmune diseases, must consider these physical parameters. Engineered biomaterials that mimic specific tissue mechanics or designed nanostructures could potentiate or dampen T-cell responses, offering novel ex vivo cell manufacturing platforms or in vivo therapeutic delivery strategies.
This whitepaper details recent advances in our understanding of nuclear mechanotransduction—the process by which mechanical forces are sensed at the cell surface, transmitted to the nucleus, and converted into specific transcriptional programs. This discussion is specifically framed within ongoing research into the biomechanical environment of CD4+ T-cells during immunological synapse (IS) formation. The mechanical cues presented by antigen-presenting cells (APCs) are now recognized as critical regulators of T-cell activation, differentiation, and effector function. Understanding how these extracellular forces lead to nuclear deformation and altered gene expression is pivotal for developing novel immunotherapies and modulating immune responses.
Core Mechanisms of Force Transmission to the Nucleus
Forces encountered at the T-cell IS are propagated through the cytoskeleton via several interconnected pathways.
- Actin-Myosin & LINC Complex Pathway: Retrograde actin flow, powered by myosin II, transmits tension from the synaptic membrane to the nucleus. This force is channeled through the Linker of Nucleoskeleton and Cytoskeleton (LINC) complex, composed of SUN and KASH domain proteins (e.g., Nesprins), which spans the nuclear envelope and connects the cytoskeleton to the nuclear lamina.
- Microtubule and Dynein Pathway: Dynamic microtubules, with the motor protein dynein, also contribute to nuclear positioning and deformation by pulling on the LINC complex.
- Nuclear Envelope and Lamina Deformation: The transmitted forces cause physical deformation of the nuclear envelope, leading to stretching and compression of the underlying nuclear lamina (a meshwork of A- and B-type lamins). This deformation alters the spacing of chromatin and lamin-associated domains (LADs).
Nuclear Mechanosensors and Transcriptional Outcomes
Nuclear deformation directly impacts chromatin organization and the activity of mechanosensitive transcriptional regulators.
- Lamin A/C as a Mechanostat: The expression level of lamin A/C correlates with nuclear stiffness. T-cells, which have relatively low lamin A/C, possess highly deformable nuclei, facilitating migration. At the IS, local lamin A/C phosphorylation and depolymerization may allow for targeted chromatin reorganization.
- YAP/TAZ Signaling: While canonical Hippo pathway signaling is important, in T-cells, YAP/TAZ localization and activity are strongly influenced by cytoskeletal tension and nuclear shape. Force-induced nuclear flattening may promote YAP/TAZ entry and co-activation of T-cell transcription factors like NFAT.
- Chromatin Remodeling via Nuclear Deformation: Mechanical strain can directly stretch chromatin fibers, reducing the density of histone marks and increasing DNA accessibility for transcription factors. This is particularly relevant for force-induced genes like EGR1 in lymphocytes.
- Emerging Role of Nuclear Pore Complexes (NPCs): Recent studies suggest NPCs may act as mechanosensors, with mechanical strain altering their conformation and affecting the selective import of transcription factors (e.g., NF-κB) into the nucleus.
Table 1: Key Transcriptional Outcomes of Nuclear Mechanotransduction in Immune Cells
| Mechanosensitive Factor | Mechanical Input | Transcriptional Outcome | Functional Implication in CD4+ T-cells |
|---|---|---|---|
| Chromatin Accessibility | Nuclear stretching/compression | Altered accessibility at specific loci (e.g., EGR1, c-FOS) | Rapid early activation and cytokine gene induction. |
| YAP/TAZ | High cytoskeletal tension, F-actin polymerization | Nuclear translocation, co-activation with TEAD/NFAT. | Promotes Th1/Th17 differentiation; regulates T-cell activation threshold. |
| NFAT | Calcium flux (downstream of TCR/pMHC kinetics) | Nuclear import and DNA binding. | Central driver of IL-2, IFN-γ production and T-cell proliferation. |
| NF-κB | LFA-1/ICAM-1 mediated adhesion and force | IκB degradation, nuclear import. | Regulates pro-survival and inflammatory genes. |
| β-Catenin | Forces on Cadherins, Wnt signaling | Nuclear translocation, complex formation with TCF/LEF. | Involved in T-cell development and memory formation. |
Experimental Protocols for Studying Nuclear Mechanotransduction
Protocol: Quantifying Nuclear Deformation in Live T-cells during Immunological Synapse Formation
Objective: To measure real-time changes in nuclear shape and volume in response to APC engagement.
- Cell Preparation: Isolate primary murine or human CD4+ T-cells. Transduce with a lentivirus expressing a nuclear localization signal (NLS)-tagged fluorescent protein (e.g., H2B-GFP).
- Substrate Preparation: Use functionalized polyacrylamide (PA) gels of tunable stiffness (0.5 - 50 kPa) coated with anti-CD3/CD28 antibodies or ICAM-1 to mimic the APC surface. For more physiological models, use supported lipid bilayers (SLBs) presenting pMHC and ICAM-1.
- Imaging: Use confocal or lattice light-sheet microscopy with environmental control (37°C, 5% CO2). Acquire z-stacks of the T-cell every 30-60 seconds for 30 minutes post-contact.
- Analysis: Segment the nucleus in 3D using software (e.g., Imaris, FIJI). Quantify metrics: Nuclear Volume (µm³), Nuclear Aspect Ratio (major/minor axis), and Nuclear Envelope Curvature.
Protocol: Assessing Force-Dependent Chromatin Accessibility (ATAC-seq on Mechanically-Stimulated T-cells)
Objective: To map genome-wide changes in chromatin architecture following specific mechanical stimulation.
- Stimulation: Stimulate purified CD4+ T-cells on functionalized PDMS micropost arrays (of defined stiffness and ligand density) or using optical tweezers to apply precise piconewton (pN) forces to surface-bound beads conjugated to anti-CD3.
- Cell Lysis & Tagmentation: At defined timepoints (e.g., 15, 60 min), lyse cells and immediately perform tagmentation using the Th5 transposase (ATAC-seq protocol). This cuts open chromatin regions.
- Library Prep & Sequencing: Amplify and sequence the tagmented DNA on a high-throughput sequencer (e.g., Illumina NovaSeq).
- Bioinformatics: Align sequences to the reference genome. Identify peaks of accessibility. Compare peaks between force-stimulated and static control cells to identify Mechanically Responsive Accessible Regions (MRARs).
Visualizing Signaling Pathways and Workflows
Diagram 1: Force Transmission from Synapse to Transcription (84 chars)
Diagram 2: Nuclear Deformation Assay Workflow (48 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Nuclear Mechanotransduction Research
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| Polyacrylamide (PA) or PDMS Hydrogels | Sigma-Aldrich, MilliporeSigma, Cytoskeleton Inc. | Fabricate substrates with physiologically tunable stiffness (0.5-50 kPa) to mimic varying tissue/APC mechanical environments. |
| Functionalization Ligands (anti-CD3ε, anti-CD28, ICAM-1-Fc) | BioLegend, R&D Systems, Sino Biological | Coat substrates to provide specific biochemical T-cell receptor and co-stimulatory signals alongside mechanical cues. |
| Lamin A/C siRNA/shRNA | Dharmacon, Santa Cruz Biotechnology | Knockdown lamin A/C to investigate its role as a nuclear mechanostat and its effect on chromatin organization and signaling. |
| YAP/TAZ Inhibitor (e.g., Verteporfin) | Selleckchem, Tocris | Chemically inhibit YAP/TAZ-TEAD interaction to delineate their role in mechano-dependent T-cell differentiation. |
| Fluorescent Nuclear Tags (H2B-GFP/RFP, NLS-Dendra2) | Addgene, MBL International | Live-cell tracking of nuclear morphology and localization of nuclear import. |
| ATAC-seq Kit | 10x Genomics, Illumina (Nextera) | Profile genome-wide chromatin accessibility changes in response to mechanical stimulation. |
| LINC Complex Antibodies (anti-SUN2, anti-Nesprin-3) | Abcam, Proteintech | Detect and localize key force-transmitting complexes at the nuclear envelope via immunofluorescence/Western blot. |
| TRACER (Tension Sensing) Reagents | As reported in literature; custom synthesis. | Genetically encoded or chemically conjugated tension sensors that report piconewton-scale forces across specific molecules (e.g., TCR, LFA-1). |
Tools of the Trade: Techniques to Probe and Perturb Synaptic Mechanics
This technical guide details the application of Traction Force Microscopy (TFM) and Micropost Array Detectors (mPADs) for quantifying cellular forces, specifically framed within ongoing research into the mechanical environment of the CD4+ T-cell immunological synapse. The formation of a stable synapse between a T-cell and an antigen-presenting cell (APC) is not merely a biochemical event but is critically modulated by mechanical forces. These forces influence receptor-ligand interactions, signaling cascades, and ultimately, T-cell activation and effector functions. Precise quantification of these piconewton- to nanonewton-scale forces is therefore essential for a complete understanding of immune cell communication and for identifying potential mechano-therapeutic targets in drug development.
Core Principles and Comparison of Techniques
Both TFM and mPADs are designed to measure tractions exerted by adherent cells on their substrate, but they operate on different physical principles.
Traction Force Microscopy (TFM): Cells are plated on a continuous, elastic hydrogel (typically polyacrylamide, PAA) embedded with fluorescent marker beads. As the cell contracts and exerts traction forces, it displaces the gel and the embedded beads. The cell is then removed (e.g., via trypsinization), allowing the gel to relax to its neutral state. Traction forces are calculated by comparing the bead displacement field between the loaded (cell-present) and reference (cell-free) states and solving an inverse mechanics problem, often using Fourier Transform Traction Cytometry (FTTC).
Micropost Arrays (mPADs): Cells are plated on a bed of flexible, vertical polydimethylsiloxane (PDMS) microposts of known geometry and spring constant. Each post acts as an independent force sensor. As the cell exerts force on the tip of a post, it deflects. The force is calculated directly using Hooke's Law (F = kδ), where k is the post's spring constant (determined by its geometry and Young's modulus) and δ is the measured deflection.
Table 1: Quantitative Comparison of TFM and Micropost Arrays
| Feature | Traction Force Microscopy (TFM) | Micropost Array Detectors (mPADs) |
|---|---|---|
| Substrate | Continuous elastic hydrogel (e.g., PAA) | Discrete PDMS microposts |
| Force Calculation | Inverse problem from displacement field (computationally intense) | Direct Hookean spring law (F = kδ) |
| Spatial Resolution | High (continuous field) | Limited to post spacing (typically 2-10 µm) |
| Force Sensitivity | ~1-10 Pa (traction stress) | ~0.1-10 nN (force per post) |
| Throughput | Lower (image processing complex) | Higher (deflection analysis is simpler) |
| Key Advantage | Continuous force map, high spatial detail | Direct, absolute force measurement, tunable stiffness |
| Main Challenge | Ill-posed inverse problem, requires reference image | Fabrication complexity, non-physiological discrete contact |
Experimental Protocols for T-Cell Immunological Synapse Studies
Protocol 3.1: Fabrication of Functionalized Polyacrylamide Gels for TFM
- Substrate Preparation: Activate glass-bottom dishes with bind-silane (3-aminopropyltrimethoxysilane) to promote gel adhesion.
- Gel Polymerization: Mix acrylamide and bis-acrylamide solutions to achieve the desired Young's modulus (e.g., 5-15 kPa for T-cells). Add fluorescent carboxylate-modified beads (0.2 µm diameter) and ammonium persulfate (APS) as an initiator.
- Surface Functionalization: Treat the polymerized gel surface with Sulfo-SANPAH under UV light to create reactive groups. Coat with extracellular matrix (ECM) proteins (e.g., fibronectin, 10 µg/mL) or, for synapse studies, with stimulatory ligands (e.g., anti-CD3ε and ICAM-1, 5 µg/mL each).
- T-Cell Plating: Isolate primary human or mouse CD4+ T-cells. Allow to settle on the functionalized gel in appropriate imaging medium. For synapse formation, use supported lipid bilayers (SLBs) presenting antigens on the gel surface as an alternative to direct protein coating.
Protocol 3.2: Traction Force Calculation via FTTC
- Image Acquisition: Acquire high-resolution fluorescence images (z-stack) of beads with the cell present.
- Reference Image: Remove the cell carefully using a gentle stream of buffer or trypsin, and acquire an image of the relaxed beads.
- Displacement Field Calculation: Use particle image velocimetry (PIV) or single-particle tracking to compute the 2D displacement vector field (u(x,y)) for all bead positions.
- Fourier Transform Traction Cytometry (FTTC): Perform the calculation in the Fourier domain. The traction field T is related to displacement u by the Green's function G of the elastic medium. The solution is regularized to avoid noise amplification.
- Metrics: Calculate total force magnitude, strain energy, and visualize traction stress vectors overlaid on cell morphology.
Protocol 3.3: Using Micropost Arrays for T-Cell Force Measurement
- Array Fabrication/Procurement: Create PDMS microposts via soft lithography from a silicon master mold, or procure commercial arrays. Posts are typically 2-10 µm in diameter and height, with center-to-center spacing of 4-15 µm.
- Surface Functionalization: Treat post tips with oxygen plasma, then incubate with fibronectin or stimulatory antibodies (anti-CD3/CD28).
- Calibration: Determine spring constant k for each post batch: k = (3πED^4)/(64L^3), where E is PDMS Young's modulus, D is post diameter, and L is height. Validate with known forces.
- Imaging and Analysis: Acquire time-lapse differential interference contrast (DIC) or fluorescence images. Track the post top and base positions. Calculate deflection δ and force F per post. Map force distribution under the cell.
TFM Experimental and Computational Workflow
Key Pathways in T-Cell Mechanotransduction
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for TFM/mPAD in T-Cell Research
| Item | Function/Benefit | Example/Notes |
|---|---|---|
| Polyacrylamide (PAA) Kit | Forms the tunable elastic gel for TFM. | CytoSoft PA Gels or in-house mix of acrylamide/bis-acrylamide. |
| Fluorescent Microbeads (200 nm) | Fiducial markers for displacement tracking in TFM. | Crimson or dark red fluorescent (far-red) beads minimize cell interference. |
| Sulfo-SANPAH Crosslinker | Activates PAA gel surface for covalent protein coupling. | Essential for stable ligand presentation under force. |
| Recombinant Proteins/Antibodies | Functionalize substrate for T-cell engagement. | Anti-CD3ε (OKT3 clone), ICAM-1-Fc, pMHC monomers. |
| PDMS (Sylgard 184) | Material for fabricating micropost arrays. | Base:curing agent ratio (e.g., 10:1) controls stiffness. |
| Calibration Cantilever | Validates force calculations for both TFM and mPADs. | Glass micro-needles or AFM cantilevers of known spring constant. |
| Live-Cell Imaging Dyes | Visualize cell structure alongside force measurement. | CellTracker dyes, F-actin labels (SiR-actin), calcium indicators (Fluo-4). |
| FTTC Analysis Software | Open-source tools for traction force calculation. | OpenTFM, PyTFM, or MATLAB-based codes (e.g., from Dr. Micah Dembo's lab). |
Data Interpretation and Application in Drug Development
Quantitative data from TFM/mPAD experiments in the CD4+ T-cell context can include: peak traction stress (Pa), total contractile moment (nN·µm), and the spatial coordination of forces relative to the synapse center.
Table 3: Example Quantitative Findings from T-Cell Studies
| Experimental Condition | Measured Force/Stress (Typical Range) | Biological Implication |
|---|---|---|
| Naïve CD4+ T-cell on anti-CD3/ICAM-1 | 1 - 5 nN total force; 50 - 200 Pa stress. | Basal level of mechanosensing during initial engagement. |
| T-cell with antigenic stimulus | 5 - 20 nN total force; 200 - 1000 Pa stress. | Force potentiation correlates with activation strength. |
| T-cell with Myosin II inhibitor (Blebbistatin) | Force reduced by 60-80%. | Confirms actomyosin dependency of traction generation. |
| T-cell on supra-physiological stiffness (>50 kPa) | Increased sustained force and altered YAP localization. | Demonstrates environmental mechanosensing impacting signaling. |
| T-cell with integrin blockade (anti-LFA1) | Reduced force transmission, unstable synapse. | Highlights integrin's role in mechanical coupling. |
For drug development professionals, these techniques offer a platform for screening immunomodulatory drugs that may act via mechanical pathways. A drug candidate that normalizes excessive T-cell force generation in autoimmune contexts, or augments it in cancer immunotherapy, represents a novel therapeutic axis. mPADs, in particular, offer a higher-throughput format for such pharmacological screening due to their direct and simpler readout.
This whitepaper provides an in-depth technical guide to two pivotal force spectroscopy techniques—Atomic Force Microscopy (AFM) and Optical Tweezers—for measuring piconewton-scale forces in single-molecule interactions. The content is specifically framed within the context of research on the mechanical microenvironment of CD4+ T-cells, particularly at the immunological synapse (IS). Understanding the forces governing T-cell receptor (TCR) engagement, integrin binding, and subsequent signaling is crucial for deciphering immune activation and for the rational design of immunotherapies and drugs modulating immune responses.
Core Principles and Instrumentation
Atomic Force Microscopy (AFM)
AFM measures force by detecting the deflection of a flexible cantilever with a sharp tip. In force spectroscopy mode, the tip is functionalized with a molecule (e.g., pMHC) and brought into contact with a cell surface molecule (e.g., TCR). As the tip retracts, unbinding events are recorded as sudden drops in force, generating a force-distance curve.
Key Parameters:
- Force Resolution: ~1-10 pN
- Spatial Resolution: Sub-nanometer (vertical), nanometer (lateral)
- Temporal Resolution: ~1-1000 ms per curve
Optical Tweezers
Optical tweezers use a highly focused laser beam to create a gradient force trap, capturing dielectric beads (typically 0.5-5 µm). By tethering a molecule between two beads or a bead and a surface, the displacement of the bead within the trap is measured to calculate applied force.
Key Parameters:
- Force Range: 0.1 - 100 pN
- Spatial Resolution: Sub-nanometer
- Temporal Resolution: Microsecond to millisecond
Table 1: Comparison of AFM and Optical Tweezers for Single-Molecule Biophysics
| Feature | Atomic Force Microscopy (AFM) | Optical Tweezers |
|---|---|---|
| Force Range | 10 pN - 10 nN | 0.1 pN - 100 pN |
| Spatial Resolution | ~0.5 nm (Z), ~1 nm (X-Y) | <1 nm (bead position) |
| Typical Loading Rates | 10 - 10^6 pN/s | 0.1 - 10^4 pN/s |
| Sample Environment | Liquid, air, vacuum; excellent for living cells | Primarily liquid; cell compatibility possible |
| Throughput | Low to medium (sequential curves) | Medium (can trap multiple beads) |
| Key Advantage | High force range, direct imaging capability, works on fixed surfaces | Superior force sensitivity & temporal resolution, passive force clamp |
| Key Limitation | Lower temporal resolution, potential tip contamination | Lower force ceiling, complex calibration in complex media |
Table 2: Measured Forces in CD4+ T-Cell Immunological Synapse Interactions
| Interacting Molecule Pair | Technique Used | Measured Unbinding Force (pN) | Loading Rate (pN/s) | Biological Context | Reference (Recent) |
|---|---|---|---|---|---|
| TCR / pMHC | Optical Tweezers | 12 - 19 | 100 - 500 | Initial antigen recognition | [1] |
| LFA-1 / ICAM-1 | AFM | 40 - 100 | 1000 - 10,000 | Adhesion & synapse stabilization | [2] |
| CD2 / CD58 | AFM | 20 - 50 | 1000 | Co-stimulation | [3] |
| PD-1 / PD-L1 | AFM | 20 - 40 | 500 - 5000 | Inhibitory checkpoint force | [4] |
Detailed Experimental Protocols
Protocol 4.1: AFM Single-Molecule Force Spectroscopy on Live T-Cells
Objective: Measure the unbinding force between a TCR and its cognate pMHC ligand.
Materials: See "The Scientist's Toolkit" below. Method:
- Cantilever Functionalization: Clean silicon nitride cantilevers (k=0.01-0.06 N/m) in piranha solution. Incubate with PEG linker containing NHS ester and biotin termini. Subsequently, incubate with streptavidin, followed by biotinylated pMHC monomer.
- Sample Preparation: Isolate primary human CD4+ T-cells or use a T-cell line. Allow cells to adhere gently to a poly-L-lysine coated glass-bottom dish in imaging buffer (e.g., HBSS with 10 mM HEPES).
- AFM Setup: Mount the dish on the AFM stage. Approach the functionalized cantilever to a cell at the periphery or over the presumed synapse region.
- Force Curve Acquisition: Set parameters (approach/retract speed: 0.5-1 µm/s, contact force: 50-100 pN, contact time: 0.1-1 s). Acquire 1000-2000 force curves at different locations.
- Data Analysis: Use algorithms (e.g., Worm-like Chain model fitting) to identify specific unbinding events from retraction curves. Plot force histograms to determine most probable unbinding force.
Protocol 4.2: Dual-Beam Optical Trap for TCR-pMHC Bond Kinetics
Objective: Measure the dissociation kinetics and force-dependent lifetime of a single TCR-pMHC bond.
Materials: See "The Scientist's Toolkit" below. Method:
- Bead and Chamber Preparation: Coat 2.1 µm streptavidin-polystyrene beads with biotinylated pMHC. Coat a second batch of beads with anti-CD3ε (to bind TCR). Alternatively, use a T-cell directly.
- Optical Trap Setup: Align two independent, overlapping laser traps in a microfluidic flow chamber.
- Molecule Tethering: Capture one bead in each optical trap. Flow in a solution containing a long, flexible dsDNA handle (~1 kb) with digoxigenin and biotin ends. Attach one end to the anti-CD3 bead via an anti-digoxigenin bridge and the other end to the pMHC bead via streptavidin-biotin.
- Force Clamp Experiment: Move one trap relative to the other to apply a constant, low force (e.g., 5-20 pN) to the tether, maintaining the bond under tension.
- Lifetime Measurement: Record the time until bond rupture (sudden bead displacement). Repeat hundreds of times to build survival probability distributions at different constant forces.
- Data Analysis: Fit data to models like the Bell-Evans model to extract zero-force off-rate (k_off) and transition state distance (Δx).
Visualization of Signaling and Workflows
Diagram 1 Title: AFM Workflow & Mechanical Signaling at Synapse
Diagram 2 Title: Optical Trap Single-Bond Kinetics Assay
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Single-Molecule Force Studies in Immunology
| Item | Function | Example/Supplier (Illustrative) |
|---|---|---|
| Biofunctionalized AFM Cantilevers | Precise force sensing with specific molecular tips. | BL-TR-TL (BioLever) from Olympus, or custom PEG-biotin-streptavidin-pMHC functionalization. |
| Long, Flexible Linkers (PEG, dsDNA) | Spacer molecules to ensure single-molecule binding and reduce non-specific adhesion. | Heterobifunctional PEG (NHS-Biotin) from Creative PEGWorks; λ-phage DNA. |
| Recombinant pMHC Monomers (Biotinylated) | High-purity, correctly folded antigen presentation complex for tip or bead coating. | NIH Tetramer Core Facility; commercial suppliers like Immudex. |
| Optical Grade Polystyrene Beads | Dielectric particles for trapping; surface chemistry for protein conjugation. | Streptavidin-coated beads (2.1 µm) from Spherotech or Polysciences. |
| Microfluidic Flow Chambers | Controlled environment for sample mounting and buffer exchange during experiments. | Custom-built using glass coverslips and double-sided tape, or commercial cells from Ibidi. |
| T-Cell Specific Media & Buffers | Maintain cell viability and physiological conditions during force measurements. | RPMI 1640 (imaging-modified), HEPES-buffered saline, 2% FBS, no phenol red. |
| Anti-CD3 / Anti-CD28 Coated Beads | For T-cell activation controls or as functionalization targets. | Dynabeads Human T-Activator CD3/CD28 from Gibco. |
| Fluorescent Tagged Antibodies (e.g., anti-CD4, anti-LFA-1) | For correlative fluorescence microscopy to identify synapse location. | Fluorochrome-conjugated antibodies from BioLegend or BD Biosciences. |
The mechanical and topographical features of the antigen-presenting cell (APC) surface are critical determinants of T-cell activation. Within the broader thesis investigating the CD4 T-cell mechanical environment in immunological synapse (IS) formation, this whitepaper details how engineered hydrogels and micropatterning serve as indispensable tools. These synthetic systems precisely control ligand presentation, substrate stiffness, and spatial organization, enabling the dissection of biophysical cues that govern T-cell receptor (TCR) signaling, cytoskeletal remodeling, and ultimate functional outcomes.
Core Principles: Decoupling Biophysical Cues
The immune synapse is a mechanosensitive structure. Traditional cell-based or rigid 2D systems conflate biochemical and physical signals. Engineered platforms allow independent tuning of:
- Ligand Density and Mobility: Controlled presentation of pMHC, adhesion molecules (e.g., ICAM-1).
- Substrate Elasticity (Stiffness): Mimicking the physiological range from soft lymphoid tissues to stiff inflamed sites (0.1 kPa to >100 kPa).
- Spatial Patterning: Dictating the geometry and arrangement of ligands to probe symmetry, clustering, and signal integration.
Material Systems: Hydrogels and Patterning Techniques
Tunable Synthetic Hydrogels
Hydrogels provide a three-dimensional, hydrous microenvironment with definable mechanical properties.
Key Materials:
- Polyacrylamide (PA): The gold standard for 2D stiffness tuning. Elastic modulus is controlled by the ratio of acrylamide to bis-acrylamide crosslinker.
- Polyethylene Glycol (PEG): Bio-inert and functionalizable via acrylate or maleimide groups. Stiffness controlled by molecular weight and crosslink density.
- Alginate: Ionically crosslinked (Ca²⁺), allowing dynamic stiffness modulation.
Quantitative Control Parameters: Table 1: Hydrogel Systems for T-Cell Mechanobiology
| Hydrogel Type | Modulus Range (kPa) | Functionalization Method | Ligand Mobility | Key Advantage |
|---|---|---|---|---|
| Polyacrylamide (PA) | 0.1 - 100 | Sulfo-SANPAH crosslinker to conjugate amines | Immobile | Precise, stable stiffness; excellent for 2D traction force microscopy. |
| PEG-Diacrylate (PEGDA) | 0.5 - 500 | Acrylate groups copolymerized with ligand-conjugated acrylates | Tunable (via PEG spacer length) | Low protein adsorption; high degree of biochemical control. |
| Alginate | 1 - 50 | Adhesion peptides (RGD) coupled via carbodiimide chemistry | Immobile | Dynamic stiffness possible; suitable for 3D encapsulation studies. |
Micropatterning Techniques
These methods create spatially defined ligand islands to control synapse architecture.
- Microcontact Printing (μCP): A PDMS stamp, inked with protein solutions, prints patterns (e.g., dots, lines) onto hydrogel or glass surfaces.
- Photopatterning: Used with photosensitive hydrogels (e.g., PEG-Norrish type I photoinitiator). UV light passed through a photomask deprotects or crosslinks specific regions for subsequent protein attachment.
- Direct Lithography: Electron-beam or dip-pen nanolithography for nanoscale patterning of ligands.
Key Experimental Protocols
Protocol 1: Fabrication of Stiffness-Tuned Polyacrylamide Hydrogels for T-Cell Stimulation
Objective: To create hydrogels of defined elasticity presenting immobilized anti-CD3/CD28 or pMHC/ICAM-1.
Materials:
- Acrylamide solution (40%), Bis-acrylamide solution (2%), PBS, TEMED, Ammonium persulfate (APS).
- Sulfo-SANPAH (ProteoChem).
- Glass coverslips activated with Bind-Silane (3-Aminopropyltriethoxysilane).
- Desired proteins: Recombinant ICAM-1, anti-CD3ε (OKT3), pMHC-II.
Method:
- Coverslip Preparation: Clean glass coverslips. Treat with Bind-Silane to promote hydrogel adhesion.
- Gel Solution Preparation: Mix acrylamide and bis-acrylamide in PBS to desired final concentrations (e.g., 5% acrylamide, 0.03% bis for ~1 kPa). Keep on ice.
- Polymerization: Add 1/100 volume each of APS (10%) and TEMED to the mix. Immediately pipette onto a silanized coverslip. Quickly place a second, untreated coverslip on top to create a thin gel sandwich.
- Gelation: Allow to polymerize for 30-45 min at room temperature.
- Functionalization: Carefully remove top coverslip. Wash gel with HEPES buffer (pH 8.5). Add Sulfo-SANPAH solution (0.5 mg/mL in HEPES) and expose to UV light (365 nm) for 5 min. Wash thoroughly.
- Protein Conjugation: Incubate gel surface with protein solution (e.g., 10 µg/mL ICAM-1 + 5 µg/mL anti-CD3) overnight at 4°C. Block with 1% BSA.
- Cell Assay: Seed isolated CD4+ T-cells onto the functionalized gel. Perform live imaging (calcium flux, tyrosine phosphorylation reporters) or fix for immunofluorescence (F-actin, pLAT, PKCθ recruitment).
Protocol 2: Microcontact Printing of Subsynaptic Ligand Patterns
Objective: To create discrete 2 μm diameter islands of stimulatory ligands surrounded by adhesion ligand alone.
Materials:
- PDMS (Sylgard 184), Silicon master wafer with positive relief features.
- Fluorescently labeled ICAM-1 (Alexa Fluor 647), anti-CD3 (Alexa Fluor 555).
- Pluronic F-127 for blocking non-patterned areas.
Method:
- Stamp Fabrication: Pour PDMS over silicon master, cure at 65°C for 2 hrs. Peel off and cut stamps.
- Inking: Incubate stamp with "inking solution" (ICAM-1 + anti-CD3 in PBS) for 1 hr in a humid chamber.
- Printing: Gently dry stamp, then bring into conformal contact with a clean, PEGylated glass surface for 30 sec.
- Blocking: Incubate printed surface with Pluronic F-127 (1% w/v) for 30 min to prevent protein adsorption in non-printed areas.
- Validation: Image patterns via fluorescence microscopy to confirm island size, density, and ligand segregation.
- T-Cell Stimulation: Seed T-cells onto patterned surface. Analyze synapse confinement to the patterned islands, and quantify signaling molecule polarization via confocal microscopy.
Signaling Pathways in a Mechanosensitive Immunological Synapse
The integration of biochemical and mechanical signals occurs through force-sensitive pathways.
Diagram 1: Key signaling pathways in T-cell mechanotransduction.
Experimental Workflow for Mechano-Immunological Studies
Diagram 2: Workflow for T-cell studies on engineered surfaces.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Hydrogel and Micropatterning Studies
| Reagent/Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Acrylamide/Bis-Acrylamide Kit | Bio-Rad, Sigma-Aldrich | Forms the backbone of tunable polyacrylamide hydrogels. |
| Sulfo-SANPAH | ProteoChem, Thermo Fisher | Heterobifunctional crosslinker for conjugating amine-containing proteins to PA gels. |
| PEG-Diacrylate (PEGDA, MW 3400-6000) | Sigma-Aldrich, Laysan Bio | Forms bio-inert, photopolymerizable hydrogels with controllable stiffness. |
| Sylgard 184 PDMS Kit | Dow, Ellsworth Adhesives | Fabrication of stamps for microcontact printing. |
| Recombinant ICAM-1 (CD54), Fc chimera | R&D Systems, Sino Biological | Key adhesion ligand for LFA-1; essential for synapse stability. |
| pMHC-II Tetramers (e.g., MCC/I-Ek) | NIH Tetramer Core, MBL International | Antigen-specific stimulation for primary CD4+ T-cells. |
| Anti-CD3ε (OKT3), functional grade | BioLegend, Thermo Fisher | Immobilized surrogate antigen for polyclonal T-cell activation. |
| Cell Tracker Dyes (CMFDA, CTV) | Thermo Fisher | Pre-labeling T-cells for live tracking and segmentation. |
| Phospho-specific Antibodies (pZAP70, pLAT) | Cell Signaling Technology | Readout of early TCR signaling events via IF. |
| Fluorophore-conjugated Phalloidin | Cytoskeleton, Inc., Thermo Fisher | Stains F-actin to visualize cytoskeletal organization in the synapse. |
This whitepaper details advanced live-cell imaging techniques for visualizing cytoskeletal dynamics, with a specific application within a broader thesis investigating the mechanical environment of CD4 T-cells during immunological synapse (IS) formation. The IS is a highly structured, dynamic interface between a T-cell and an antigen-presenting cell (APC), where mechanical forces and cytoskeletal rearrangements are critical for signal transduction, effector function, and fate decisions. Understanding the real-time spatial and temporal orchestration of actin, microtubules, and associated proteins at the IS requires cutting-edge imaging methodologies.
Core Imaging Modalities and Quantitative Comparison
High-resolution live-cell imaging of cytoskeletal dynamics employs several complementary modalities. The table below summarizes their key characteristics, performance data, and suitability for studying T-cell mechanics.
Table 1: Comparison of High-Resolution Live-Cell Imaging Modalities
| Modality | Spatial Resolution (XY) | Temporal Resolution | Key Advantage for Cytoskeleton/IS | Primary Limitation |
|---|---|---|---|---|
| TIRF Microscopy | ~100 nm | Millisecond to second | Excellent signal-to-noise for cortical actin at synapse; minimal photobleaching. | Images only ~100-200 nm from coverslip. |
| Spinning Disk Confocal | ~200-250 nm | Sub-second to seconds | Good optical sectioning for 3D dynamics in the cell body. | Lower light throughput vs. TIRF. |
| Lattice Light-Sheet (LLS) | ~200 nm (XY) ~300 nm (Z) | Sub-second | Extremely low phototoxicity for long-term 4D (x,y,z,t) imaging of whole cell. | Complex setup; sample mounting. |
| STED Super-Resolution | ~30-80 nm | Seconds to minutes | True super-resolution for nanoscale actin organization. | High light intensity; slower imaging. |
| SIM Super-Resolution | ~100 nm | Seconds | Good resolution improvement for dynamic microtubule networks. | Reconstruction artifacts possible. |
Recent data (2023-2024) indicates that for IS studies, TIRF remains the workhorse for synapse-proximal actin dynamics, with LLS adoption growing for holistic 3D force and cytoskeleton coupling studies. A hybrid TIRF/LLS system can achieve ~150 nm XY resolution at 0.5-2 second intervals, enabling concurrent synaptic and volumetric imaging.
Detailed Experimental Protocol: Imaging Actin Dynamics at the T-cell Immunological Synapse
This protocol describes the process for visualizing actin flow in primary human CD4 T-cells during synapse formation with supported lipid bilayers (SLBs) presenting antigen.
Protocol 3.1: Cell Preparation and Labeling
- Isolate CD4+ T-cells: Isolate naive CD4 T-cells from human peripheral blood using a negative selection kit. Activate with plate-bound anti-CD3/anti-CD28 for 3-4 days, then rest in IL-2.
- Transduce with Biosensor: Transduce activated T-cells with a lentivirus encoding Lifeact-mNeonGreen (for F-actin) or EGFP-β-actin. Use a low MOI to ensure moderate expression. Sort for positive cells 48-72 hours post-transduction.
- Alternative Labeling: For non-transduced cells, use a cell-permeable fluorogenic actin probe (e.g., SiR-actin) at 100-500 nM for 1 hour prior to imaging.
Protocol 3.2: Supported Lipid Bilayer (SLB) Formation
- Prepare glass-bottom dishes (e.g., 35mm, No. 1.5 coverslip) by plasma cleaning.
- Form small unilamellar vesicles (SUVs) from a lipid mix containing DOPC, DOGS-NTA(Ni), and biotinylated-cap-DPPE (molar ratio 97:2:1).
- Fuse SUVs onto the cleaned glass to form a continuous bilayer.
- Load the bilayer with recombinant His-tagged ICAM-1, and streptavidin-tagged antigenic pMHC complexes. Incubate at 37°C for 30 min.
Protocol 3.3: TIRF Microscopy Imaging Setup
- Microscope: Use a TIRF microscope equipped with a 100x/1.49 NA oil immersion TIRF objective, 488 nm laser, and an EM-CCD or sCMOS camera.
- Environmental Control: Maintain stage at 37°C using a heater and use a perfusion chamber with 5% CO₂.
- Image Acquisition:
- Add labeled T-cells (1-2 x 10⁵ cells/mL) in imaging medium to the SLB chamber.
- Set the TIRF angle to achieve an evanescent field depth of ~150 nm.
- Acquire time-lapse images with an exposure time of 50-100 ms at 1-2 second intervals for 10-15 minutes.
- Maintain laser power below 0.5-1 kW/cm² to minimize phototoxicity.
Protocol 3.4: Data Analysis (Kymograph Generation)
- Define Line of Interest: Draw a straight line across the immunological synapse, from the distal to the proximal pole.
- Generate Kymograph: Use Fiji/ImageJ (Multi Kymograph plugin) to create a space-time plot.
- Quantify Flow: Measure the slope of diagonal streaks in the kymograph to calculate actin retrograde flow velocity (typically 0.1 - 0.3 µm/sec in primary T-cells).
Key Signaling Pathways Visualized
Cytoskeletal dynamics at the IS are governed by intricate signaling pathways triggered by TCR-pMHC engagement. The diagram below outlines the core pathway leading to actin polymerization and remodeling.
TCR to Actin Polymerization Signaling Cascade
Integrated Workflow for IS Cytoskeleton Analysis
The experimental journey from hypothesis to quantitative data involves a multi-step workflow, integrating biology, imaging physics, and computational analysis.
IS Cytoskeleton Imaging and Analysis Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Live Imaging of T-cell Cytoskeletal Dynamics
| Reagent / Material | Category | Function & Rationale | Example Product / Target |
|---|---|---|---|
| Fluorescent Actin Biosensors | Live-cell probes | Label F-actin with minimal perturbation for long-term imaging. | Lifeact-EGFP/mNeonGreen, F-tractin-tdTomato, SiR-actin (chemical). |
| Microtubule Probes | Live-cell probes | Visualize microtubule dynamics and centrosome reorientation. | EGFP-EMTB, mRuby-TUBB (tubulin), SiR-tubulin. |
| Supported Lipid Bilayer (SLB) Kits | Synthetic substrate | Present antigen and adhesion molecules in a controllable, fluid 2D membrane to trigger IS formation. | Cytiva's Bio-sup-ported membranes or custom DOPC/DOGS-NTA formulations. |
| Myosin Inhibitors | Pharmacological agents | Probe the role of contractility in cytoskeletal dynamics and force generation. | Blebbistatin (Myosin II), ML-7 (MYLK). |
| ROCK / Formin Inhibitors | Pharmacological agents | Dissect specific pathways of actin polymerization (formin) and regulation (ROCK). | SMIFH2 (Formin), Y-27632 (ROCK). |
| High-Performance Imaging Media | Cell culture media | Maintain cell viability and function during imaging, minimal fluorescence background. | FluoroBrite DMEM, CO₂-independent medium with HEPES and supplements. |
| Glass-Bottom Dishes (No. 1.5) | Imaging hardware | Provide optimal optical clarity and correct thickness for high-NA objectives. | MatTek dishes, CellVis imaging dishes. |
| Anti-Fade Reagents (for fixed validation) | Imaging enhancers | Preserve fluorescence in fixed samples for correlative super-resolution studies. | ProLong Diamond, SlowFade Glass. |
High-resolution live-cell imaging is indispensable for dissecting the spatiotemporal mechanics of the T-cell immunological synapse. The integration of TIRF, LLS, and super-resolution modalities with robust protocols and quantitative analysis, as outlined here, provides a powerful framework for testing hypotheses within a broader thesis on CD4 T-cell mechanical signaling. Future advancements in adaptive optics, lower-toxicity biosensors, and AI-driven image analysis will further unlock the ability to visualize and quantify the molecular mechanics of immunity in real time.
This whitepaper explores the frontier of immunotherapy drug discovery focused on the mechanobiology of the immune synapse. The central thesis is that the mechanical forces exerted and sensed by CD4+ T cells during antigen presentation are critical regulators of immune activation and function. These mechanosensitive pathways present a novel class of therapeutic targets for modulating immune responses in cancer, autoimmunity, and chronic inflammation. Targeting the proteins and signaling cascades that translate mechanical cues into biochemical signals offers a paradigm shift from traditional biochemical-centric approaches.
The Mechanobiology of the CD4+ T Cell Immunological Synapse
The immunological synapse (IS) is a highly organized, dynamic interface between a T cell and an antigen-presenting cell (APC). Beyond biochemical signaling, it is a site of significant mechanical force generation and sensing.
Key Mechanical Processes:
- TCR Triggering: The T-cell receptor (TCR) engages peptide-MHC (pMHC) on the APC. The bond is subjected to piconewton (pN)-scale forces, which can modulate TCR signaling kinetics and affinity discrimination—a process termed mechanosensing.
- Actin Cytoskeleton Remodeling: The integrin LFA-1 binds to ICAM-1 on the APC, forming the peripheral supramolecular activation cluster (pSMAC). Actin retrograde flow, driven by myosin IIA, generates sustained tension critical for signal amplification and sustained signaling.
- Nuclear Translocation of Transcription Factors: Forces transmitted via the linker of nucleoskeleton and cytoskeleton (LINC) complex can influence chromatin remodeling and gene expression, linking immediate mechanical events to long-term transcriptional programs.
Core Mechanosensitive Pathways and Druggable Targets
The following pathways translate mechanical stimuli into biochemical signals. Their components are high-value targets for drug discovery.
| Pathway Name | Core Mechanosensor(s) | Key Downstream Effectors | Primary Cellular Outcome | Druggability Assessment |
|---|---|---|---|---|
| TCR-PKCθ Mechanotransduction | TCR-pMHC bond, Talin, Kindlin | PLC-γ1, PKCθ, CARMA1, NF-κB | T cell activation, IL-2 production | High (Kinase inhibitors, allosteric modulators) |
| Integrin (LFA-1) Force Signaling | LFA-1-ICAM-1 bond, Talin, Vinculin | RhoA/ROCK, FAK, PI3K | Adhesion strengthening, cytoskeletal polarization, costimulation | Medium-High (Small molecule antagonists, conformational lockers) |
| YAP/TAZ Hippo Pathway | Actin cytoskeleton tension, LINC complex | YAP, TAZ, TEADs | Proliferation, survival, differentiation (Th1/Th17) | High (TEAD inhibitors, YAP/TAZ-TEAD disruptors) |
| PIEZO1 Channel Flux | PIEZO1 ion channel (membrane tension) | Ca²⁺ influx, NFAT, Calcineurin | Activation, cytokine profiling | Medium (Channel blockers, activators) |
Experimental Protocols for Mechano-Immunology Research
Quantifying TCR-pMHC Binding Forces (Single-Molecule Force Spectroscopy)
This protocol uses an Atomic Force Microscope (AFM) functionalized with recombinant pMHC to measure the unbinding force from TCR on a live T cell.
- Functionalization: Cantilevers are coated with PEG linker and biotin, followed by streptavidin and biotinylated pMHC.
- Cell Preparation: Primary human CD4+ T cells are activated for 48-72 hours and adhered to a poly-L-lysine-coated dish in serum-free media.
- Force Measurement: The pMHC-coated tip is brought into contact with the T cell surface (100 pN contact force, 0.5-1 sec dwell time). The tip is then retracted at a constant velocity (500-1000 nm/s).
- Data Analysis: The rupture force (pN) is extracted from the retraction curve. Thousands of measurements are compiled into a force histogram to determine the characteristic unbinding force.
Imaging Cytoskeletal Forces in the Immunological Synapse (Traction Force Microscopy)
This protocol maps the forces a T cell exerts on its substrate during antigen recognition.
- Substrate Fabrication: Prepare polyacrylamide gels (elastic modulus ~10 kPa, mimicking APC stiffness) embedded with 0.2 µm fluorescent beads.
- Surface Coating: Coat gel surface with anti-CD3ε (simulates APC) and ICAM-1-Fc.
- Cell Seeding & Imaging: Allow primary CD4+ T cells to settle on the substrate. Acquire time-lapse images (confocal microscope) of bead displacements.
- Force Calculation: After removing the cell (trypsin), image the bead reference position. Use particle image velocimetry (PIV) algorithms to calculate bead displacement vectors and convert to traction stress (Pa) using the gel's known mechanical properties.
Assessing YAP/TAZ Nuclear Translocation (A Readout of Mechanical Signaling)
A simple immunofluorescence assay to measure the activity of the mechanosensitive Hippo pathway.
- Stimulation: Seed CD4+ T cells on stimulatory surfaces of varying stiffness (soft: ~1 kPa, stiff: ~50 kPa) coated with anti-CD3/CD28.
- Fixation & Permeabilization: At desired timepoints (e.g., 24h), fix cells with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 10 min.
- Staining: Block with BSA, incubate with anti-YAP/TAZ primary antibody overnight at 4°C, then with fluorescent secondary antibody for 1h. Include DAPI for nuclei.
- Quantification: Image with a fluorescence microscope. Calculate the nuclear-to-cytoplasmic fluorescence intensity ratio (N/C ratio) for YAP/TAZ for ≥100 cells per condition using image analysis software (e.g., ImageJ).
Visualization of Key Signaling Pathways
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Category | Specific Example(s) | Function in Mechanosensitive Research |
|---|---|---|
| Functionalizable Surfaces | Polyacrylamide hydrogels of tunable stiffness (e.g., Softwell plates); Supported lipid bilayers (SLBs) with mobile pMHC/ICAM-1. | Provides a biomimetic, mechanically defined substrate for T cell stimulation, enabling isolation of stiffness as a variable. |
| Recombinant Proteins | Biotinylated pMHC monomers/tetramers; Fc-chimeric ICAM-1, CD80; His-tagged antigens. | Essential for functionalizing AFM tips, gels, or SLBs for controlled mechanical engagement of TCRs and integrins. |
| Mechanosensor Modulators | YAP/TAZ inhibitor Verteporfin; PIEZO1 agonist Yoda1; ROCK inhibitor Y-27632; Myosin II inhibitor Blebbistatin. | Pharmacological tools to perturb specific nodes in mechanotransduction pathways and assess functional outcomes. |
| Force-Activated Probes | DNA-based tension gauge tethers (TGTs); Fluorescent FRET-based molecular tension sensors (e.g., TCR or integrin-specific). | Reporters that visualize and quantify piconewton-scale forces across specific receptor-ligand pairs in live cells. |
| Actin/LFA-1 Labels | Phalloidin (F-actin stain); Anti-LFA-1 (CD11a) antibody for imaging; LifeAct-GFP transfected cell lines. | Visualize cytoskeletal architecture and adhesion molecule organization in the synapse in response to mechanical cues. |
| Advanced Imaging Systems | Total Internal Reflection Fluorescence (TIRF) microscope; Atomic Force Microscope (AFM) with fluid cell; Confocal microscope with live-cell chamber. | Enables high-resolution, real-time visualization and physical measurement of synaptic dynamics and forces. |
Navigating Experimental Pitfalls: Optimization Strategies for Mechanobiology Assays
Common Artifacts in 2D vs. 3D Culture Systems and How to Mitigate Them
The study of CD4 T-cell activation, particularly the formation and function of the immunological synapse (IS), is heavily influenced by the mechanical and spatial environment. Traditional 2D culture systems, while foundational, introduce artifacts that can misrepresent in vivo T-cell biology. This guide contrasts common artifacts in 2D versus 3D systems within the context of CD4 T-cell mechanical environment research and provides mitigation strategies.
Core Artifacts: A Quantitative Comparison
Table 1: Key Artifacts in 2D vs. 3D CD4 T-Cell Synapse Studies
| Artifact Category | 2D System Manifestation | 3D System Manifestation | Impact on IS/Mechanics Research |
|---|---|---|---|
| Substrate Rigidity | Non-physiological, uniform high rigidity (e.g., ~1 GPa glass). | Tunable, more physiological (e.g., 0.1-10 kPa hydrogels). | 2D overemphasizes cytoskeletal forces and alters TCR microcluster dynamics. |
| Ligand Presentation | Non-physiological density & distribution (fixed, planar). | More native mobility and clustering (on beads or in matrix). | 2D forces sustained central supramolecular activation cluster (cSMAC) not always seen in vivo. |
| Cell Morphology & Polarity | Strongly adherent, exaggerated spreading, fixed polarity. | Dynamic, amoeboid, multi-polar engagements possible. | 2D over-simplifies kinapse vs. synapse behavior and signal integration. |
| Diffusion & Soluble Factors | Gradients are difficult to establish; paracrine signaling is limited. | Stable chemokine/cytokine gradients form; autocrine/paracrine effects are pronounced. | 2D misrepresents chemotaxis and sustained activation thresholds. |
| Forces & Confinement | Lacks in vivo-like spatial confinement and force vectors. | Can incorporate physical confinement and 3D force application. | 2D fails to model the role of mechanical confinement in IS maturation. |
Table 2: Mitigation Strategies & Experimental Outcomes
| Strategy | Protocol/Method | Key Parameter Changed | Outcome on IS Metrics (Typical Data Range) |
|---|---|---|---|
| Polyacrylamide Hydrogels | Functionalize with ICAM-1 & anti-CD3ε. Tune crosslinker:bis ratio. | Substrate Elasticity (0.5 - 20 kPa) | Peak F-actin polarization shifts from <5 kPa. Calcium flux optimal at ~1-3 kPa. |
| 3D Collagen Matrices | Embed T-cells & antigen-presenting cells (APCs) in high-density rat-tail collagen I (3-5 mg/mL). | Pore Size & Ligand Mobility | Synapse lifetime decreases (2D: >30 min; 3D: ~10-15 min). Multi-polar engagements increase to ~30%. |
| Magnetic Twisting | Coat beads with stimulatory ligands. Apply oscillatory torque via magnetic field. | Applied Shear Stress (0.1 - 10 pN/μm²) | TCR triggering threshold increases with force. PKCθ translocation enhanced at 2-5 pN/μm². |
| Microfluidic Gradients | Generate stable CXCL12 or CCL19 gradients in a 3D microchannel. | Chemokine Gradient (0-100 ng/mL over 200 μm) | Directed migration speed increases from ~5 μm/min (2D) to ~12 μm/min (3D). |
Detailed Experimental Protocols
Protocol 1: Fabricating Tunable Stiffness 2D Hydrogels for IS Imaging
- Surface Activation: Clean glass coverslips with NaOH and treat with Bind-Silane (0.5% v/v).
- Gel Solution: Mix 40% acrylamide and 2% bis-acrylamide stocks in PBS to desired stiffness (e.g., 3 kPa: 5% acrylamide, 0.15% bis). Add 0.1% TEMED and 0.5% ammonium persulfate (APS) to initiate polymerization.
- Functionalization: Incubate polymerized gels with Sulfo-SANPAH (0.2 mg/mL) under UV light, then coat with recombinant ICAM-1 (5 μg/mL) and anti-CD3ε antibody (2 μg/mL) overnight at 4°C.
- Cell Seeding: Plate purified primary CD4 T-cells and allow synapse formation (37°C, 5% CO₂) for 10-20 minutes before fixation and staining.
Protocol 2: 3D Collagen Matrix Synapse Assay
- Matrix Preparation: On ice, mix high-concentration rat-tail collagen I (8-10 mg/mL) with 10X PBS, 0.1M NaOH for pH adjustment, and complete cell culture medium to achieve a final 4 mg/mL collagen, pH 7.4.
- Cell Embedding: Resuspend pre-labeled CD4 T-cells and antigen-loaded B-cell blasts or dendritic cells in the cold collagen solution at 1-2 x 10⁶ cells/mL each.
- Polymerization: Quickly pipette 50 μL drops into μ-Slide 3D culture chambers. Incubate at 37°C for 30 minutes to polymerize.
- Culture & Imaging: Add warm medium overlay. Image live using confocal microscopy within 2-6 hours to capture IS dynamics in 3D.
Visualizing the Experimental Workflow & Signaling
Diagram 1: Workflow for Addressing 2D Artifacts in IS Research
Diagram 2: Mechanical Environment Modulates Synapse Signaling
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Mechano-Immunological Synapse Studies
| Item | Function & Relevance | Example Product/Catalog |
|---|---|---|
| Tunable Hydrogel Kit | Provides substrates of defined elasticity (0.5-50 kPa) to isolate rigidity effects on IS formation. | Cytosoft Rigidity Tuning Plates, Polyacrylamide Gel Kits. |
| 3D ECM Hydrogel | Creates a physiological 3D scaffold for studying cell migration and synapse formation in confinement. | Corning Matrigel, Rat Tail Collagen I, High Concentration. |
| Functionalized Beads | Enable precise control of ligand density and type for reductionist IS stimulation under force. | Streptavidin Polystyrene Beads (for biotinylated pMHC/antibodies). |
| Magnetic Tweezers/Cell Strainer | Applies quantified physical force to receptor-ligand bonds to study mechanotransduction. | Magnetic Twisting Cytometry systems, Flexcell Tension systems. |
| Microfluidic Gradient Generator | Creates stable, defined soluble gradients to study chemokine-guided synapse polarity in 3D. | Ibidi µ-Slide Chemotaxis, Elveflow OB1 pressure controller. |
| Live-Cell Imaging Dyes | For real-time visualization of calcium flux, actin dynamics, and synaptic architecture. | Fluo-4 AM (Calcium), SiR-Actin (F-actin), CellTracker dyes. |
| pMHC Tetramers/Multimers | Specifically label and stimulate antigen-specific T-cells for physiologically relevant studies. | NIH Tetramer Core, MBL International custom pMHC complexes. |
The activation and function of CD4+ T-cells are profoundly influenced by the mechanical properties of their microenvironment. During immunological synapse (IS) formation with antigen-presenting cells (APCs), T-cells probe and respond to stiffness cues, which modulate receptor clustering, signaling cascade initiation, and ultimate effector functions. The extracellular matrix (ECM) stiffness in lymphoid organs and peripheral tissues varies from soft (healthy parenchyma) to stiff (fibrotic or tumor microenvironments). This technical guide provides a framework for calibrating hydrogel systems to mimic these physiological (~0.1 - 10 kPa) and pathological (up to 50+ kPa) stiffness ranges, ensuring biological relevance for studies of T-cell mechanobiology.
Defining the Stiffness Landscape for CD4+ T-Cell Research
Physiological and Pathological Stiffness Ranges
Quantitative data on tissue stiffness relevant to CD4+ T-cell biology is compiled from recent literature.
Table 1: Stiffness of T-Cell Microenvironments
| Tissue / Microenvironment | Elastic Modulus (kPa) | Pathological State | Measurement Technique |
|---|---|---|---|
| Bone Marrow | 0.3 - 0.6 | Normal | Atomic Force Microscopy (AFM) |
| Lymph Node Cortex | 0.2 - 2 | Normal | Micropipette Aspiration |
| Lung Parenchyma | 0.5 - 1.5 | Normal | AFM |
| Healthy Liver | 0.4 - 1 | Normal | Shear Wave Elastography |
| Fibrotic Liver | 8 - 25 | Pathological | Shear Wave Elastography |
| Breast Tissue | 0.2 - 0.4 | Normal | AFM |
| Breast Tumor (Invasive Ductal Carcinoma) | 1 - 15+ | Pathological | AFM, Macro-indentation |
| Atherosclerotic Plaque | 20 - 100 | Pathological | AFM |
Impact of Stiffness on CD4+ T-Cell Function
Key functional outcomes modulated by substrate stiffness include:
- Proliferation: Optimal on intermediate stiffness (~5-10 kPa), often suppressed on very soft or very stiff substrates.
- Cytokine Polarization: Stiffer substrates tend to promote Th1/Th17 profiles, while softer substrates may favor regulatory T-cell (Treg) development.
- Cytoskeletal Organization: Increased stiffness promotes F-actin polarization and denser actin arcs at the IS.
- Mechanosensing: Engaged through proteins like Talin, Vinculin, and the mechanosensitive ion channel Piezo1.
Core Hydrogel Systems for Stiffness Tuning
Table 2: Common Hydrogel Platforms for T-Cell Mechanobiology
| Hydrogel Base | Crosslinking Mechanism | Typical Stiffness Range | Key Advantages | Considerations for T-Cell Work |
|---|---|---|---|---|
| Polyacrylamide (PA) | Free-radical polymerization (Bis-acrylamide crosslinker) | 0.1 - 50 kPa | Decoupled biochemical/mechanical cues; well-established. | Requires surface functionalization (e.g., collagen, ICAM-1) for T-cell adhesion. |
| Polydimethylsiloxane (PDMS) | Polymer base-to-curing agent ratio | 10 kPa - 3 MPa | High optical clarity; easy to pattern. | Stiffness range often supra-physiological; absorbs small hydrophobic molecules. |
| Polyethylene Glycol (PEG) | Michael-type addition, photopolymerization | 0.1 - 100 kPa | High bio-inertness; precise biochemical functionalization. | Requires incorporation of adhesive ligands (RGD, anti-CD3/CD28). |
| Alginate | Ionic (Ca²⁺) crosslinking | 0.5 - 100 kPa | Injectable; adaptable to 3D culture. | Batch variability; ligand coupling can be complex. |
| Fibrin/Collagen I (Natural) | Enzymatic/physical self-assembly | 0.01 - 5 kPa | Naturally bioactive; inherent cell adhesion sites. | Biochemical composition co-varies with stiffness. |
Detailed Experimental Protocols
Protocol: Fabricating Polyacrylamide Hydrogels of Tunable Stiffness for T-Cell Plating
This protocol generates hydrogel-coated coverslips with defined elastic moduli.
Reagents & Materials:
- 40% Acrylamide stock solution
- 2% Bis-acrylamide stock solution
- 1 M HEPES buffer, pH 8.5
- Ammonium persulfate (APS) solution (10% w/v, fresh)
- Tetramethylethylenediamine (TEMED)
- Sulfo-SANPAH (in DMSO)
- Functionalization ligand (e.g., Recombinant ICAM-1, Collagen I)
- 18 mm or 25 mm glass coverslips, activated with Bind-Silane
- PBS (sterile)
Procedure:
- Coverslip Activation: Clean coverslips with ethanol and treat with 0.5% (v/v) Bind-Silane (3-aminopropyltrimethoxysilane) in acetone for 5 minutes. Rinse and dry.
- Hydrogel Solution Preparation: For desired stiffness, mix acrylamide and bis-acrylamide stocks in dH₂O according to Table 3. Add HEPES to a final concentration of 0.1 M.
- Polymerization: Degas mixture for 10 min. Add 1/100 volume of 10% APS and 1/1000 volume TEMED. Mix and immediately pipet 15-20 µL onto a Bind-Silane treated coverslip. Quickly place a clean, untreated coverslip on top to form a sandwich.
- Curing: Allow to polymerize for 30-45 minutes at room temperature.
- Lifting & Washing: Gently separate the sandwich with a razor blade. Wash hydrogel-coated coverslips in PBS.
- Surface Functionalization:
- Place gels under UV lamp for 5 minutes.
- Incubate with 0.5 mg/mL Sulfo-SANPAH in PBS under UV for 10 minutes.
- Wash 3x with PBS.
- Incubate with functionalization ligand (e.g., 5 µg/mL ICAM-1 in PBS) overnight at 4°C.
- Wash and store in PBS at 4°C for up to 1 week.
Table 3: Polyacrylamide Formulations for Target Stiffness
| Target Elastic Modulus (kPa) | % Acrylamide (Final) | % Bis-acrylamide (Final) | Young's Modulus (Approx., kPa)* |
|---|---|---|---|
| 0.5 | 3 | 0.03 | 0.3 - 0.7 |
| 2 | 5 | 0.06 | 1.5 - 2.5 |
| 8 (Physiological Lymph Node) | 7.5 | 0.15 | 7 - 9 |
| 20 (Fibrotic/Tumor) | 10 | 0.3 | 18 - 25 |
| 50 (Severe Pathology) | 12 | 0.5 | 45 - 60 |
*As verified by AFM.
Protocol: Atomic Force Microscopy (AFM) for Hydrogel Stiffness Validation
Direct mechanical measurement is critical for quality control.
Procedure:
- Probe Selection: Use a spherical tip probe (5-10 µm diameter) for soft materials.
- Calibration: Perform thermal tune in air to determine spring constant (typically 0.01-0.1 N/m for soft gels).
- Measurement: Approach hydrogel surface in PBS at room temperature. Obtain force-indentation curves at ≥10 random locations per gel.
- Analysis: Fit the retraction curve segment to the Hertz contact model for a spherical indenter to calculate the Young's Elastic Modulus (E).
Key Signaling Pathways in T-Cell Mechanotransduction
Diagram Title: T-Cell Mechanosensing Pathways at the IS
Experimental Workflow for Stiffness-Dependent T-Cell Assays
Diagram Title: Workflow for Stiffness-Based T-Cell Experiments
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Reagent Solutions for Hydrogel-Based T-Cell Mechanobiology
| Reagent / Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Acrylamide/Bis-acrylamide Kit | Bio-Rad, Sigma-Aldrich | Forms the backbone of tunable PA hydrogels. |
| Sulfo-SANPAH | Thermo Fisher Scientific | Heterobifunctional crosslinker for covalently attaching proteins to PA gel surface. |
| Recombinant Human ICAM-1/Fc Chimera | R&D Systems | Critical adhesion ligand for LFA-1 on T-cells; enables IS formation on synthetic gels. |
| Anti-CD3ε (OKT3) & Anti-CD28 (CD28.2) | BioLegend, Tonbo | For functionalization onto gels or beads to provide TCR/CD28 stimulation. |
| Polydimethylsiloxane (PDMS) Sylgard 184 Kit | Dow Chemical | For fabricating stiff (>50 kPa) or micropatterned substrates. |
| 4-Arm PEG-Acrylate (PEG-4A) | Laysan Bio, Sigma | Building block for inert, tunable PEG hydrogels via photopolymerization. |
| Y-27632 (ROCK inhibitor) | Tocris Bioscience | Inhibits actomyosin contractility; used as a control to disrupt force generation. |
| GsMTx-4 Peptide | Alomone Labs | Selective inhibitor of Piezo1 mechanosensitive channels. |
| Fluorescent Beads (0.5-2 µm) | Polysciences, Invitrogen | For traction force microscopy (TFM) to measure T-cell contractile forces. |
| Atomic Force Microscope (MFP-3D, BioFastScan) | Asylum Research, Bruker | Gold-standard instrument for direct nanomechanical measurement of hydrogel stiffness. |
Faithful recapitulation of the in vivo mechanical landscape is non-negotiable for elucidating the role of stiffness in CD4+ T-cell activation at the immunological synapse. By rigorously calibrating hydrogel systems using the formulations, validation protocols, and functionalization strategies outlined herein, researchers can generate physiologically relevant data. This approach will accelerate the discovery of mechanobiological targets in immune dysfunction and pave the way for novel therapeutic strategies in fibrosis, cancer, and autoimmune diseases.
Optimizing Ligand Density and Mobility to Isolate Mechanical Cues
Within the broader thesis investigating how the mechanical environment of the CD4 T-cell immunological synapse (IS) dictates functional outcomes, this guide details the precise manipulation of ligand presentation. By systematically controlling the surface density and lateral mobility of peptide-MHC (pMHC) and co-stimulatory ligands (e.g., ICAM-1), researchers can isolate and quantify mechanical cues—such as ligand rigidity, force transmission, and spatial patterning—independent of biochemical signaling variance. This technical whitepaper provides a roadmap for experimental design, protocol implementation, and data interpretation to mechanistically dissect T-cell mechanosensing.
The formation of the IS is not merely a biochemical event but a biomechanical process. CD4 T-cells dynamically probe the antigen-presenting cell (APC) surface, generating and responding to physical forces. These mechanical cues, integrated with TCR-pMHC binding kinetics, modulate T-cell activation, differentiation, and cytokine polarization. Isolating mechanical contributions requires engineering substrates where biochemical ligand identity and concentration are held constant while parameters like ligand density (molecules/µm²) and mobility (diffusion coefficient) are precisely varied. This approach directly tests hypotheses central to the overarching thesis on the role of the mechanical microenvironment in adaptive immunity.
Foundational Concepts and Quantitative Benchmarks
Table 1: Key Ligand Parameters and Their Mechanical Impact
| Parameter | Definition | Experimental Range (Typical) | Primary Mechanical Consequence | Measurable T-cell Output |
|---|---|---|---|---|
| Surface Density | Number of ligand molecules per unit area. | pMHC: 0.1 - 100 molecules/µm²; ICAM-1: 50 - 1000 molecules/µm² | Modulates bond cluster size, lateral tension, and spatial organization of force application. | Actin retrograde flow rate, traction force magnitude, synapse stability. |
| Lateral Mobility | Diffusion coefficient (D) of ligand in membrane or supported lipid bilayer (SLB). | D in SLB: 0.1 - 5.0 µm²/s; Tethered: D ≈ 0 µm²/s | Determines capacity for ligand rearrangement under force, enabling or resisting cytoskeletal coupling. | Centripetal movement of ligands (kinapse vs. synapse), phosphorylation kinetics of mechanosensors (e.g., CasL). |
| Substrate Elasticity/ Rigidity | Young's modulus of the presenting surface. | 0.1 kPa (soft) to 100+ kPa (stiff) | Governs overall deformability, influencing the magnitude of reaction forces generated by T-cell pulling. | Nuclear localization of mechanotransduction factors (YAP/TAZ), proliferation depth. |
| Ligand Tethering | Covalent vs. fluid linkage to substrate. | Flexible PEG tethers (length: 2-40 nm), direct covalent fixation. | Controls the length scale over which force can be exerted before ligand detachment, acting as a mechanical "gauge". | Sustained calcium signaling, integrin activation thresholds. |
Core Experimental Methodologies
Fabrication of Tunable Ligand Surfaces
Protocol 1: Creating Supported Lipid Bilayers (SLBs) with Defined Mobility and Density
- Objective: To present fluid pMHC and ICAM-1 ligands where density and mobility are independently controlled.
- Materials: DOPC, DOGS-NTA(Ni), biotinylated-cap-DPPE lipids, purified His-tagged pMHC, streptavidin, biotinylated ICAM-1.
- Steps:
- Prepare small unilamellar vesicles (SUVs) by extrusion. Lipid composition dictates mobility: high DOPC (>99%) yields high fluidity; adding cholesterol increases viscosity; incorporating biotinylated lipids provides docking points.
- Form an SLB on a clean glass coverslip in a flow chamber by incubating with SUV solution, followed by extensive washing.
- Control Density: For pMHC, inject His-tagged protein at varying concentrations; density is controlled by the mol% of Ni-NTA lipids and incubation time/concentration. For ICAM-1, inject streptavidin, then biotinylated ICAM-1 at defined concentrations.
- Quantify: Use fluorescence recovery after photobleaching (FRAP) to measure diffusion coefficient (mobility). Use calibrated total internal reflection fluorescence (TIRF) microscopy or quantitative fluorescence to determine absolute density (molecules/µm²).
Protocol 2: Fabrication of Non-Fluid, Micropatterned Surfaces
- Objective: To present immobile ligands at precisely defined spatial geometries and densities.
- Materials: Glass substrates, PEG-based resist, deep UV lamp, micro-contact stamp, functionalized with pMHC/ICAM-1.
- Steps:
- Use deep UV photolithography or micro-contact printing to create adhesive protein patterns (e.g., 2-5 µm dots, lines) on a non-adhesive PEG background.
- Backfill the PEG area with passivating protein (e.g., BSA).
- Incubate patterned substrate with precise concentrations of pMHC and ICAM-1 to achieve specific density via adsorption kinetics.
- Key Control: Verify ligand immobility via FRAP and pattern fidelity via high-resolution microscopy.
Critical Measurement Techniques
Protocol 3: Traction Force Microscopy (TFM) on Tunable Substrates
- Objective: To measure the spatiotemporal forces exerted by a T-cell on engineered surfaces.
- Materials: Polyacrylamide gel (varying stiffness, e.g., 1-50 kPa) embedded with 0.2 µm fluorescent beads, functionalized with sulfo-SANPAH for protein conjugation.
- Steps:
- Covalently couple ligands (pMHC/ICAM-1) to the gel surface at defined densities.
- Seed primary CD4 T-cells or T-cell lines onto the functionalized gel.
- Acquire time-lapse images of beads during IS formation.
- Calculate displacement fields from bead movement and compute traction stresses using Fourier Transform Traction Cytometry algorithms.
Protocol 4: Quantifying Mechanosensing via Fluorescent Biosensors
- Objective: To visualize early mechanotransduction events in live T-cells.
- Materials: T-cells expressing FRET-based tension sensors (e.g., TSMod inserted into TCR-CD3ζ or CD28 cytoplasmic domains) or localization reporters (e.g., YAP-GFP).
- Steps:
- Activate sensor-expressing T-cells on surfaces with varying ligand mobility/density.
- Image using TIRF or confocal microscopy to measure FRET efficiency (indicative of molecular tension) or nuclear/cytoplasmic translocation of YAP/TAZ.
- Correlate sensor readouts with ligand presentation parameters.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Isolating Mechanical Cues
| Item / Reagent | Function in Experiment | Example / Key Consideration |
|---|---|---|
| His-tagged pMHC Monomers | Presents antigenic peptide to TCR. Allows controlled density via Ni-NTA chemistry. | Recombinant mouse I-Aᵇ loaded with OVA³²³⁻³³⁹ peptide. Critical: Ensure peptide loading efficiency >95%. |
| Biotinylated ICAM-1 | Presents LFA-1 ligand for adhesion and co-stimulation. Density controlled via streptavidin-biotin. | Recombinant mouse ICAM-1-Fc with AviTag. Use monomeric streptavidin to prevent cross-linking. |
| DOPC & Functionalized Lipids | Forms the basis of fluid SLBs. DOGS-NTA(Ni) and biotin-cap-DPPE allow specific ligand attachment. | Avanti Polar Lipids catalog #850375, #790404, #870273. Maintain high purity (>99%). |
| PEG-Based Passivation Reagents | Creates non-fouling, inert backgrounds on solid substrates to isolate patterned signals. | PEG-Silane for glass; PLPP-PEG-biotin for blocking and functionalization. |
| Polyacrylamide Gel Kit | Creates substrates of tunable elasticity for TFM and rigidity sensing studies. | Cytoskeleton, Inc. Traction Force Kit, or prepare from acrylamide/bis-acrylamide stocks. |
| Fluorescent Fiducial Beads | Embedded in gels for TFM to visualize substrate deformation. | Crimson FluoSpheres (0.2 µm, 645/680 nm). Choose wavelength far from cell labels. |
| FRET-based Tension Sensor (TSMod) | Genetically encoded reporter for molecular-scale force across specific proteins. | Insert into CD3ζ or CD28 cytoplasmic domains to measure TCR/LFA-1 engagement forces. |
| Anti-phospho Specific Antibodies | Fixed-cell readout of mechanosensitive pathway activation. | Anti-pY165 CasL, anti-pY397 FAK. Use for immunofluorescence after timed engagements. |
Signaling Pathways in T-cell Mechanotransduction
Diagram Title: T-cell Mechanotransduction Pathway from Engineered Ligands
Integrated Experimental Workflow
Diagram Title: Workflow for Isolating Mechanical Cues in T-cell Studies
Data Interpretation and Integration into the Broader Thesis
Interpreting data from these experiments requires multi-parametric analysis. For example, a finding that high pMHC density on a fluid SLB fails to trigger robust YAP nuclear translocation, while the same density on a rigid, immobile substrate does, isolates the critical role of ligand immobilization and substrate deformability in activating the Hippo pathway. This directly tests the thesis postulate that mechanical memory is encoded via cytoskeletal remodeling sustained by resistive force. Data should be structured to compare outputs (e.g., traction stress maps, phospho-signal intensities) across a matrix of ligand density (X-axis) and mobility (Y-axis) for each substrate rigidity.
Conclusion: The precise optimization of ligand density and mobility is a foundational methodology for deconvoluting mechanical cues in immunological synapse research. The protocols and frameworks provided here enable direct testing of hypotheses concerning how physical forces, distinct from biochemical signals, guide CD4 T-cell fate decisions—a core objective of the broader thesis on the T-cell mechanical microenvironment.
Within the context of CD4 T-cell immunological synapse research, a central challenge is the accurate interpretation of cellular responses that result from integrated chemical and mechanical signaling. The immunological synapse is a complex, dynamic interface where T-cell receptors (TCRs) engage peptide-MHC complexes on antigen-presenting cells (APCs), co-receptors bind ligands, and integrins mediate adhesion. Critically, this microenvironment presents both biochemical cues (e.g., cytokines, antigen affinity) and biophysical cues (e.g., substrate stiffness, APC membrane rigidity, topological features). Disentangling the specific contributions of each modality is essential for understanding fundamental immunology and for the rational design of immunotherapies and vaccines that may rely on engineered biomaterial scaffolds.
The Integrated Signaling Landscape
T-cell activation is not a binary switch but a tunable response governed by the summation of signals. The following table summarizes key signaling outputs and their known or potential chemical and mechanical triggers.
Table 1: T-Cell Activation Outputs and Their Putative Triggers
| Signaling Output / Phenotype | Chemical Triggers (Examples) | Mechanical Triggers (Examples) |
|---|---|---|
| Early TCR Signaling (pZAP70, pLAT) | High-affinity pMHC, CD4 engagement | Force-mediated TCR-pMHC bond stabilization, substrate stiffness |
| Calcium Influx | PLCγ1 activation via LAT | Cytoskeletal tension modulating ORAI1 channel activity |
| NFAT Nuclear Translocation | Sustained calcium flux | Actin flow dynamics regulating signalosome localization |
| Immunological Synapse Maturation | PKCθ activation, Lck signaling | Micropatterning, actomyosin-driven TCR microcluster movement |
| Cytokine Polarization (e.g., IFN-γ) | IL-12, strong TCR signal duration | 3D matrix confinement, interstitial pressure |
| Proliferation | IL-2 secretion and receptor engagement | Shear stress, stiffness-modulated survival signals |
| Metabolic Reprogramming | mTOR activation via PI3K-Akt | Matrix elasticity influencing cytoskeletal mTORC1 localization |
Methodological Framework for Disentanglement
To isolate mechanical from chemical effects, researchers employ engineered systems that permit orthogonal control of variables. The following experimental protocols represent key approaches.
Protocol 1: Tunable Hydrogel Substrates for Stickness Studies
Objective: To isolate the effect of substrate stiffness on CD4 T-cell activation independent of ligand density. Materials: Polyacrylamide hydrogels, functionalized with recombinant ICAM-1 and anti-CD3ε antibodies via sulfo-SANPAH crosslinking. Procedure:
- Prepare hydrogels of defined elastic moduli (e.g., 0.5 kPa, 10 kPa, 50 kPa) by varying acrylamide/bis-acrylamide ratios.
- Confirm stiffness using atomic force microscopy (AFM) indentation.
- Functionalize surface with a fixed, saturating density of anti-CD3ε and ICAM-1. Quantify via fluorescence calibration.
- Isolate naive CD4 T-cells from mouse spleen or human PBMCs. Allow to settle on hydrogel.
- Activate for specified time points (e.g., 5 min for early signaling, 24h for proliferation).
- Fix and stain for phosphoproteins (e.g., pZAP70), image via confocal microscopy, or analyze by flow cytometry.
- Critical Control: Perform identical experiment on rigid glass coated with the identical ligand density.
Protocol 2: DNA-Based Tension Probes (TCR-pMHC)
Objective: To directly quantify piconewton (pN) forces across individual TCR-pMHC bonds. Materials: DNA hairpin tension probes (e.g., ~12-19 pN unzipping force) conjugated to pMHC and fluorophore/quencher pair. Procedure:
- Synthesize and conjugate pMHC to the DNA tension probe. The fluorophore is quenched in the folded state.
- Pattern probes onto a passivated glass surface at low density to allow single-molecule resolution.
- Incubate with CD4 T-cell line (e.g., Jurkat) or primary T-cells.
- Image using TIRF microscopy. Fluorescence emission indicates probe unfolding due to mechanical force applied by the TCR.
- Correlate force events (fluorescence spots) with downstream calcium flux (using Fluo-4 AM dye) in the same cell.
- Key Variation: Use probes with different force thresholds to map the "force spectrum" required for activation.
Protocol 3: Optical Tweezers/Traction Force Microscopy
Objective: To apply defined mechanical loads to TCRs and measure cellular traction forces during synapse formation. Materials: Optical tweezer setup, polystyrene beads coated with anti-CD3/CD28; polyacrylamide gels with embedded fluorescent beads for traction force microscopy (TFM). Tweezer Protocol:
- Coat 1-µm beads with anti-CD3. Optically trap and position a bead against a T-cell.
- Apply oscillatory or constant displacement force.
- Monitor early signaling (e.g., calcium) via ratiometric dyes. TFM Protocol:
- Plate T-cells on anti-CD3/ICAM-1-coated polyacrylamide gel with fiducial markers.
- Track displacement of beads directly beneath the cell during activation.
- Compute traction stress fields using Fourier transform techniques.
Visualizing Signaling Pathways and Workflows
Diagram 1: Integrated Chemical-Mechanical Signaling in CD4 T-Cell Activation
Diagram 2: Workflow for Single-Molecule TCR Force Measurement
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Mechano-Immunology Studies
| Reagent/Material | Supplier Examples | Function in Disentanglement Studies |
|---|---|---|
| Polyacrylamide Hydrogel Kits | Bio-Rad, Sigma-Aldrich, Cytoskeleton Inc. | Provides tunable-stiffness 2D substrates for isolating mechanical microenvironment effects. |
| Functionalized Beads (Polystyrene) | Polysciences, Spherotech | Used with optical tweezers or static presentation to apply controlled force via attached anti-CD3/CD28. |
| DNA Tension Probe Kits (pMHC) | Custom synthesis (e.g., from Lumicks) | Conjugatable probes that report piconewton-scale forces across specific receptor-ligand bonds. |
| Recombinant pMHC Monomers | NIH Tetramer Core, MBL International | Defined chemical ligands for TCRs; essential for coupling to tension probes or patterning surfaces. |
| Fluorescent Ca2+ Indicators (Fluo-4, Fura-2) | Thermo Fisher, Abcam | Ratimetric or intensity-based dyes to measure downstream signaling activation in live cells. |
| Phospho-Specific Antibodies (pZAP70, pLAT) | Cell Signaling Technology | Critical for quantifying early biochemical signaling events via flow cytometry or microscopy. |
| Microfabricated PDMS Pillars | CYTOO, custom fabrication | Arrays of flexible posts whose deflection measures cellular traction forces at sub-cellular resolution. |
| Inhibitors (Blebbistatin, Y-27632) | Tocris, Sigma | Pharmacologically disrupts actomyosin contractility to test the role of cellular force generation. |
Disentangling chemical and mechanical signaling in CD4 T-cell activation remains a formidable but essential challenge. It requires a multidisciplinary toolkit combining bioengineering, biophysics, molecular biology, and advanced microscopy. The protocols and frameworks outlined here provide a roadmap for designing experiments that yield interpretable data. As the field progresses, the integration of these orthogonal data streams will be crucial for building predictive models of immune cell function and for translating this knowledge into next-generation therapeutic strategies that intelligently modulate the immunomechanical interface.
Best Practices for Reproducible Force Measurement and Statistical Analysis
The mechanical microenvironment at the immunological synapse (IS) between a CD4+ T-cell and an antigen-presenting cell (APC) is a critical determinant of T-cell activation, differentiation, and effector function. Precise and reproducible measurement of the piconewton-scale forces generated during this dynamic interaction is foundational to a mechanistic understanding of immune signaling. This guide establishes a rigorous framework for obtaining, analyzing, and reporting force data within this specific biological context, ensuring that findings are robust, comparable, and biologically meaningful.
Core Principles of Reproducibility
Reproducibility in this field hinges on three pillars: instrumental calibration, biological standardization, and transparent statistical reporting. Variations in cell type, activation state, substrate functionalization, and environmental control (temperature, CO₂) are major sources of irreproducibility. A comprehensive laboratory log that tracks all parameters (Table 1) is non-negotiable.
Table 1: Essential Metadata for Reproducible Force Experiments
| Category | Specific Parameter | Example/Standard | Recording Frequency |
|---|---|---|---|
| Cell System | T-cell line/primary source, stimulation status, culture passage | Jurkat D1.1, primary human naïve CD4+, anti-CD3/CD28 activated for 48h | Per experiment |
| Instrument | AFM model, cantilever type & lot, spring constant calibration method | Bruker MLCT-Bio-DC, k=0.01 N/m, thermal tune method | Per cantilever use |
| Substrate | Functionalization ligand, density, coupling chemistry, passivation | anti-CD3e (OKT3), 200 molecules/μm², PEG silane passivation | Per batch |
| Environment | Buffer composition, temperature control, perfusion rate | RPMI-1640 + 10% FBS, 37°C ± 0.5°C, no flow | Continuous monitoring |
| Acquisition | Trigger force, approach/retract velocity, sampling rate | 50 pN trigger, 1 μm/s velocity, 10 kHz sampling rate | Per measurement cell |
Experimental Protocols for T-Cell Force Measurement
Protocol: Cantilever Functionalization for TCR Engagement
Objective: To coat AFM cantilevers with a recombinant protein capable of engaging the T-cell receptor (TCR) complex with specificity and controlled geometry.
- Cleaning: Plasma clean tipless cantilevers (e.g., Bruker MLCT-Bio-DC) for 5 minutes.
- PEG Spacer Attachment: Incubate in 1 mM mPEG-SCM (5 kDa) and biotin-PEG-SCM (5 kDa) (95:5 ratio) in chloroform for 2 hours. This forms a mixed monolayer.
- Washing: Rinse thoroughly in chloroform and ethanol, then dry under N₂.
- Ligand Attachment: Incubate in 0.1 mg/mL streptavidin in PBS for 10 minutes. Rinse with PBS.
- Functionalization: Incubate in 10 μg/mL biotinylated anti-CD3e Fab fragment (clone OKT3) in PBS for 30 minutes. Rinse with imaging buffer.
- Validation: Confirm functionalization via force-distance curves on a surface coated with free biotin.
Protocol: Live-Cell Force Spectroscopy on CD4+ T-Cells
Objective: To measure the engagement forces between a TCR-functionalized probe and a live CD4+ T-cell under physiological conditions.
- Cell Preparation: Allow primary human CD4+ T-cells or Jurkat cells to adhere to a poly-L-lysine-coated Petri dish in pre-warmed, CO₂-equilibrated imaging buffer (e.g., Leibovitz's L-15 + 10% FBS).
- Instrument Setup: Mount functionalized cantilever. Calibrate spring constant using the thermal noise method in situ. Set temperature to 37°C.
- Approach: Position the cantilever over the cell's central region. Set approach velocity to 0.5-1.0 μm/s with a trigger force of 30-50 pN.
- Contact & Dwell: Upon trigger, maintain contact for a defined dwell time (0.1-2.0 s) to allow synapse formation.
- Retract: Retract the cantilever at a constant velocity (0.5-1.0 μm/s) while recording deflection.
- Replication: Perform ≥ 100 force-curves per cell condition, across ≥ 3 biological replicates (different cell donors/passages).
Data Processing & Statistical Analysis Framework
Raw force-distance curves must be processed consistently:
- Baseline Correction: Subtract the non-contact linear deflection.
- Conversion to Force: Force (F) = Deflection (d) × Spring Constant (k).
- Adhesion Event Detection: Use a consistent threshold (e.g., 15 pN above baseline) to identify unbinding events.
- Feature Extraction: For each curve, extract:
- Peak Adhesion Force (Fmax): The maximum unbinding force.
- Work of Adhesion (W): The area under the retraction curve.
- Rupture Length (Lr): The distance to the final rupture event.
Table 2: Summary Statistics & Recommended Tests for Common Comparisons
| Biological Question | Primary Data | Distribution Check | Recommended Statistical Test | Effect Size Metric |
|---|---|---|---|---|
| Force change upon drug inhibition (e.g., Blebbistatin) | F_max per curve | Non-normal, positive skew | Mann-Whitney U test (non-paired) | Hodges-Lehmann median difference |
| Comparison between T-cell subtypes (e.g., naïve vs. Th17) | Median F_max per cell | Approximately normal | Unpaired t-test (with Welch's correction) | Cohen's d |
| Time-course of force after activation | Mean work of adhesion per time point | Varies | One-way ANOVA with Tukey's post-hoc | Eta-squared (η²) |
| Correlation between force and calcium flux | Paired (F_max, Ca²⁺) per cell | Bivariate | Spearman's rank correlation | Spearman's ρ |
Key Reporting Requirements: Always report the exact N (number of cells and curves), the measure of central tendency (median for non-normal data), and dispersion (interquartile range or mean ± SD). Provide data distribution plots (e.g., box plots with superimposed data points).
Signaling Pathways in Mechanotransduction at the IS
The application and detection of force at the IS initiates specific signaling cascades that modulate T-cell responses.
Experimental Workflow for Reproducible Studies
A standardized workflow from sample prep to publication is essential.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for T-Cell Force Measurement
| Item | Example Product/Catalog | Function |
|---|---|---|
| Tipless Cantilevers | Bruker MLCT-Bio-DC (k=0.01 N/m) | The force sensor; requires precise spring constant calibration. |
| Biotin-PEG Linker | Biotin-PEG-SCM, 5 kDa (Nanocs) | Creates a flexible, non-fouling tether for ligand presentation. |
| TCR Engagement Ligand | Recombinant biotinylated anti-CD3ε Fab (Clone OKT3) | Specifically engages the TCR complex without Fc receptor confounding. |
| Passivation Agent | mPEG-Silane (MW 2000-5000) | Passivates substrate surfaces to prevent non-specific adhesion. |
| Myosin Inhibitor | (-)-Blebbistatin (Sigma-Aldrich) | Specific inhibitor of non-muscle myosin II to test actomyosin dependence. |
| Calibration Grid | Bruker TGXYZ1 (or similar) | For lateral piezoelectric scanner calibration. |
| Temperature Controller | Petri dish heater with feedback (Bioscience Tools) | Maintains physiological temperature during live-cell imaging. |
| Data Processing Software | JPK Data Processing (or custom Python/R scripts) | For consistent, automated baseline correction and feature extraction. |
Integrating rigorous force measurement protocols with robust statistical practices is paramount for advancing our understanding of CD4+ T-cell mechanobiology. By adhering to the standards of instrumental calibration, biological replication, and transparent data analysis outlined herein, researchers can generate reproducible, high-quality data that reliably elucidates the force-dependent mechanisms governing the immunological synapse.
Context and Validation: Comparing CD4 Mechanobiology Across States and Subsets
This whitepaper, framed within the broader thesis of CD4 T-cell mechanical environment immunological synapse research, explores the distinct mechanical strategies employed by CD4+ helper and CD8+ cytotoxic T lymphocytes. While both cell types utilize the immunological synapse (IS) to engage antigen-presenting cells (APCs) or target cells, recent biomechanical studies reveal fundamental differences in their force application, synaptic architecture, and mechanosensing. These divergent strategies are optimized for their unique effector functions: CD4+ cells provide sustained signaling for cytokine-mediated help, while CD8+ cells deliver rapid, lethal force for target cell killing. Understanding these mechanisms provides novel targets for immunotherapies.
The immunological synapse is a dynamic, force-generating interface. The mechanical environment—encompassing stiffness, ligand mobility, and topography—is a critical regulator of T-cell signaling and function. This review synthesizes current data to argue that CD4+ and CD8+ T-cells have evolved distinct mechanical phenotypes.
Quantitative Comparison of Mechanical Properties
Table 1: Summary of Key Mechanical and Synaptic Properties of CD4+ vs. CD8+ T-Cells
| Property | CD4+ T-Cells (Helper) | CD8+ T-Cells (Cytotoxic) | Measurement Technique |
|---|---|---|---|
| Peak Traction Force | ~12-20 nN (sustained) | ~50-100 nN (peak, transient) | Traction Force Microscopy (TFM) on polyacrylamide gels |
| Synapse Stability | Prolonged (>30 min), stable | Rapid (<5 min), dynamic | Live-cell imaging, TIRF microscopy |
| Actin Flow Dynamics | Centripetal, organized, slower | Arcs, rapid, contractile bursts | FRAP, Lifeact-GFP imaging |
| Force Application Pattern | Continuous, pushing/pulling | Pulsed, highly focused on target | Atomic Force Microscopy (AFM), TFM |
| Optimal APC/Target Stiffness | Intermediate (10-30 kPa) | Broad range, effective on softer targets (≤5 kPa) | Stiffness-tunable hydrogel substrates |
| LFA-1/ICAM-1 Clutch Engagement | High-avidity, sustained | Transient, rapid engagement/disengagement | Biomembrane Force Probe (BFP) |
| Key Mechanosensor | TCR-pMHC-II bond reinforcement | Integrin-mediated stiffness sensing | Genetic knockout, FRET-based tension sensors |
| Cytoskeletal Regulator Bias | WASp/Arp2/3 (branched actin) | Myosin IIA (contractility) | Pharmacologic inhibition, siRNA |
Experimental Protocols for Key Mechanobiological Assays
Protocol 3.1: Traction Force Microscopy (TFM) for T-Cell Force Mapping
Objective: Quantify magnitude and spatial distribution of forces exerted by single T-cells on deformable substrates.
- Substrate Preparation: Fabricate polyacrylamide gels (PAA) of defined stiffness (e.g., 5, 20 kPa) embedded with 0.2 µm fluorescent microbeads (reference layer) and coated with anti-CD3ε/anti-CD28 antibodies or ICAM-1.
- Cell Loading: Isolate naïve or activated human/mouse CD4+ or CD8+ T-cells. Allow cells to settle and adhere to the functionalized gel for 2-5 minutes in imaging medium.
- Imaging: Acquire high-resolution z-stacks of bead positions (under cell) and a reference image after cell removal (using trypsin-EDTA or detergent).
- Analysis: Use particle image velocimetry (PIV) or Fourier transform traction cytometry to compute bead displacement fields. Solve the inverse Boussinesq problem to calculate traction stress vectors and total force.
Protocol 3.2: Biomembrane Force Probe (BFP) for Single-Bond Kinetics
Objective: Measure the force-dependent kinetics of TCR-pMHC and LFA-1-ICAM-1 bonds at the single-molecule level.
- Probe Assembly: A glass micropipette holds a human red blood cell (RBC) aspirated into a bullet shape. The RBC is coated with streptavidin and linked to a biotinylated glass bead, forming the force probe.
- Functionalization: The bead is coated with a single or few recombinant pMHC or ICAM-1 proteins. The T-cell is held by a separate micropipette.
- Measurement: The probe bead is brought into contact with the T-cell for a set time. Upon retraction, the RBC acts as a soft spring. Bond formation and rupture forces are measured via high-speed video analysis of bead displacement relative to the pipette.
- Data Acquisition: Record thousands of touch cycles to construct force-clamp or force-ramp curves, determining bond lifetime and catch/slip bond behavior.
Protocol 3.3: FRET-Based Tension Sensor Imaging in Live Synapses
Objective: Visualize molecular-scale tension across specific proteins (e.g., TCR, CD3, LFA-1) in real-time.
- Sensor Construction: Transduce T-cells with a lentivirus encoding the tension sensor module (TSMod). TSMod is inserted into the protein of interest (e.g., in the CD3ε cytoplasmic domain) and consists of a fluorophore pair (mTFP1 and Venus) connected by a defined elastic linker.
- Synapse Formation: Allow transduced T-cells to engage supported lipid bilayers (SLBs) presenting pMHC and ICAM-1, or target cells.
- Imaging: Perform live confocal or TIRF microscopy. FRET efficiency (mTFP1 emission / Venus emission) is inversely proportional to tension on the module.
- Analysis: Generate ratiometric FRET maps of the synapse to visualize nanonewton-scale force distribution across the engaged receptors.
Key Signaling and Mechanotransduction Pathways
Diagram Title: Divergent TCR and Mechanotransduction Pathways in CD4+ vs. CD8+ T-Cells
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for T-Cell Mechanobiology Research
| Reagent/Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Tuneable Polyacrylamide Hydrogel Kits | Cell Guidance Systems, Matrigen | Provides substrates of precise stiffness (0.1-100 kPa) for Traction Force Microscopy and stiffness-sensing studies. |
| Fluorescent Microbeads (0.2 µm) | Thermo Fisher (FluoSpheres), Phosphorex | Embedded in gels as fiduciary markers for displacement tracking in TFM. |
| Recombinant pMHC-I & pMHC-II Tetramers/Monomers | NIH Tetramer Core, MBL International, BioLegend | For antigen-specific T-cell staining, isolation, and functional studies on SLBs or AFM cantilevers. |
| Supported Lipid Bilayer (SLB) Components | Avanti Polar Lipids | DOPC, DOGS-Ni-NTA lipids for constructing fluid planar membranes presenting pMHC and ICAM-1. |
| FRET-Based Tension Sensor (TSMod) Constructs | Addgene (Plasmids from Zhu et al.) | Genetically encoded modules to visualize piconewton forces across specific cell surface receptors. |
| Myosin II Inhibitor (Blebbistatin) | Tocris, Sigma-Aldrich | Specific inhibitor to dissect the role of contractility in CD8+ T-cell killing or CD4+ synapse stability. |
| Arp2/3 Inhibitor (CK-666) | Tocris, Sigma-Aldrich | Inhibits branched actin nucleation, critical for probing actin dynamics in CD4+ T-cell synapse organization. |
| Biomembrane Force Probe (BFP) Setup | Custom fabrication (see protocol) | Enables single-molecule, force-dependent bond kinetics measurement for TCR and integrins. |
| High-Speed Live-Cell Imaging System | PerkinElmer, Nikon, Zeiss | Essential for capturing rapid cytoskeletal dynamics (actin arcs, granule movement) in cytotoxic synapses. |
The divergent mechanical strategies of CD4+ and CD8+ T-cells—optimized for sustained signaling versus explosive force delivery—represent a fundamental principle of immune system engineering. For drug development, this implies:
- Adoptive Cell Therapies: Engineering CAR-T cells may require tuning mechanical signaling pathways (e.g., modulating Lck activity or integrin engagement) to match the desired effector profile (cytokine helper vs. cytotoxic).
- Checkpoint Inhibitors: The mechanical microenvironment of tumors may differentially impede CD4+ vs. CD8+ infiltration and function. Therapies could target mechanosensing pathways (e.g., ROCK, Myosin II) to enhance T-cell penetration.
- Vaccine Design: Adjuvants and delivery systems should consider the mechanical context (e.g., lymph node stiffness, APC surface properties) to optimally prime the appropriate helper or cytotoxic response.
Future research within the thesis framework must further dissect how the CD4+ T-cell's mechanical environment dictates its ultimate functional polarization (Th1, Th2, Treg) and how dysregulation of these mechanical programs contributes to autoimmunity and immunodeficiency.
This whitepaper explores the stage-specific mechanosensitivity of CD4+ T-cell subsets within the context of immunological synapse (IS) formation and signaling. Recent advances highlight how the mechanical properties of the T-cell environment and the forces exerted during antigen recognition are differentially integrated by naive, effector, and memory T-cell subsets, leading to distinct functional outcomes. This mechanosensing capability is a critical, non-biochemical regulator of adaptive immune responses, with significant implications for therapeutic intervention.
The immunological synapse is a dynamically structured, force-generating interface between a T-cell and an antigen-presenting cell (APC). Beyond biochemical signaling, the IS is a site of active mechanical force application and sensation. T-cells probe the APC surface using actin-driven protrusions and exert pushing and pulling forces via integrins and the T-cell receptor (TCR) complex. The cellular response to these mechanical cues—including substrate stiffness, ligand mobility, and tensile forces—varies dramatically with T-cell differentiation state. This document synthesizes current research on how naive, effector, and memory T-cell subsets decode these mechanical signals.
Stage-Specific Mechanosensitive Phenotypes
Each T-cell subset exhibits a unique mechanosensitive profile, optimized for its functional role.
- Naive T-Cells: Require sustained, high-affinity pMHC engagement on a relatively stiff dendritic cell (DC) surface for full activation. They are highly sensitive to substrate stiffness, with optimal proliferation on surfaces mimicking lymphoid tissue (∼1-10 kPa). They generate moderate forces to test ligand quality.
- Effector T-Cells: Primed for rapid cytokine secretion and cytotoxicity. They are less dependent on substrate stiffness and can form functional synapses on softer target cell membranes. They exert high, sustained contractile forces via the actin-myosin machinery to facilitate killing and interrogate target cells.
- Memory T-Cells: Exhibit a hybrid phenotype. Central memory (T_CM_) cells retain some stiffness sensitivity akin to naive cells, while tissue-resident memory (T_RM_) cells adapt to the mechanics of peripheral tissues. They can re-activate quickly across a broader range of mechanical contexts.
Table 1: Comparative Mechanosensitive Properties of CD4+ T-Cell Subsets
| Property | Naive T-Cell | Effector T-Cell | Memory T-Cell |
|---|---|---|---|
| Optimal Substrate Stiffness | 1-10 kPa (Lymphoid) | 0.5-5 kPa (Broad) | Context-Dependent (0.5-10 kPa) |
| TCR Triggering Force Threshold | High (∼10-15 pN) | Lower (∼5-12 pN) | Intermediate |
| Peak Traction Force Exerted | Moderate (∼5-20 nN) | High (∼15-50 nN) | Variable (5-30 nN) |
| Actin Retrograde Flow Rate | Fast, exploratory | Slow, forceful | Adaptable |
| Primary Mechanosensitive Pathway | TCR-pLAT via Talin1 | TCR-ZAP70 via Lck | Integrated TCR & Integrin signals |
| Key Mechanosensor Molecule | Talin1, Vinculin | Lck, Myosin IIA | PIP5K1C, Kindlin-3 |
Experimental Protocols for Assessing T-Cell Mechanosensitivity
Traction Force Microscopy (TFM) on Polyacrylamide Gels
Purpose: To quantify the magnitude and spatial distribution of forces exerted by T-cells on substrates of defined stiffness.
- Substrate Preparation: Fabricate polyacrylamide (PA) gels of specific Young's modulus (e.g., 1, 5, 20 kPa) conjugated with ICAM-1 and anti-CD3ε antibodies via sulfo-SANPAH crosslinking. Embed 0.2 μm fluorescent beads as fiducial markers.
- Cell Seeding: Isolate and purify T-cell subsets (e.g., via FACS sorting for CD44, CD62L, CD45RO). Allow cells to settle onto the functionalized gel in imaging medium.
- Image Acquisition: Acquire time-lapse images of bead displacement (TxRed channel) and cell morphology (DIC or GFP) using a high-resolution confocal or TIRF microscope every 10-30 seconds for 15-30 minutes.
- Force Calculation: After cell removal (using trypsin or a detergent), acquire a reference image of relaxed beads. Use particle image velocimetry (PIV) algorithms to compute bead displacement fields. Traction stresses are calculated by solving the inverse elastic problem using Fourier Transform Traction Cytometry (FTTC).
Biomembrane Force Probe (BFP) Single-Molecule Assay
Purpose: To measure the precise force and kinetics of single TCR-pMHC bond rupture and downstream calcium signaling.
- Probe Preparation: A red blood cell (RBC) is aspirated by a micropipette to form a spring-like force probe. A glass bead coated with recombinant pMHC is attached to the RBC apex.
- T-Cell Preparation: A single T-cell is aspirated by a second micropipette. The cell is brought into contact with the pMHC bead.
- Force Measurement: Using precision piezo actuators, the T-cell is pressed against the bead to initiate bond formation and then retracted at controlled speeds. The deflection of the RBC (measured via video microscopy) directly reports the force on the bond.
- Simultaneous Ca²⁺ Imaging: T-cells are loaded with a calcium indicator (e.g., Fluo-4 AM). Fluorescence intensity is monitored during force application to correlate mechanical engagement with early signaling.
Structured Illumination Microscopy (SIM) of Actin Dynamics
Purpose: To visualize nanoscale architecture and dynamics of the actin cytoskeleton during IS formation on mechanically tuned surfaces.
- Sample Preparation: Seed T-cells onto stimulatory surfaces (functionalized glass or PA gels) in an imaging chamber.
- Staining: Fix cells at specific time points (e.g., 2, 5, 10 min post-contact) and stain for F-actin (Phalloidin), TCR, and phosphorylated proteins (e.g., pZAP70).
- Imaging: Image samples using a commercial SIM microscope with a high-NA objective. Acquire z-stacks encompassing the entire cell-substrate interface.
- Analysis: Reconstruct high-resolution 3D images. Quantify parameters such as actin clearance at the central supramolecular activation cluster (cSMAC), peripheral F-actin density, and co-localization of TCR with force-sensitive proteins like Talin.
Key Signaling Pathways in Stage-Specific Mechanotransduction
Diagram Title: Core Mechanosignaling Pathways in T-Cell Subsets
Research Reagent Solutions Toolkit
Table 2: Essential Reagents for T-Cell Mechanobiology Research
| Reagent/Category | Example Product/Description | Primary Function in Research |
|---|---|---|
| Tunable Hydrogels | Polyacrylamide (PA) gels, PDMS microposts, PEG-based hydrogels | Provide substrates with defined, physiological stiffness for 2D cell culture and force measurement. |
| Functionalization Ligands | Recombinant ICAM-1, anti-CD3ε/anti-CD28 antibodies, streptavidin for biotinylated pMHC | Covalently attach stimulatory molecules to gels or beads to mimic APC surfaces. |
| Fluorescent Force Probes | DNA-based tension gauge tethers (TGTs), FRET-based force sensors (e.g., TSMod) | Visualize and quantify molecular-scale forces across specific receptor-ligand pairs (e.g., TCR-pMHC). |
| Actin/Live-Cell Labels | SiR-actin, LifeAct-GFP, CellMask dyes | Label cytoskeleton and cell membrane for live imaging of IS dynamics without fixation. |
| Calcium Indicators | Fluo-4 AM, Fura-2 AM, GCaMP6f-expressing T-cell lines | Monitor early activation via intracellular Ca²⁺ flux in response to mechanical stimulation. |
| Mechanosensor Antibodies | Anti-phospho-ZAP70 (Tyr319), Anti-phospho-Lck (Tyr394), Anti-Talin (open conformation) | Detect force-sensitive biochemical events by flow cytometry or microscopy. |
| Inhibitors/Agonists | Blebbistatin (Myosin II inhibitor), Y-27632 (ROCK inhibitor), Mn²⁺ (Integrin activator) | Modulate specific components of the cellular force-generation machinery. |
| Microfluidic Devices | Custom chips for cell pairing, stiffness gradients, or shear stress application | Create controlled microenvironments for high-throughput mechanostimulation assays. |
The stage-specific mechanosensitivity of T-cells represents a fundamental layer of immune regulation. Naive cells use force as a stringent checkpoint for antigen quality, effector cells harness it for potent function, and memory cells employ it for contextual adaptability. Targeting these mechanobiological pathways—for instance, by modulating integrin activation or myosin contractility—offers a novel avenue for immunotherapy. Enhancing the mechanosensing of tumor-infiltrating lymphocytes or suppressing the pathological force generation in autoimmune disorders could be the next frontier in precision immunology. Future research must move towards 3D mechano-mimetic environments and in vivo force sensing to fully translate these principles.
This whitepaper examines the mechanical dimension of T-cell dysfunction in solid tumors, framed within a broader thesis on the role of the CD4+ T-cell mechanical environment in immunological synapse (IS) formation and signaling. The pathological extracellular matrix (ECM) stiffness, a hallmark of desmoplastic tumors, is not merely a structural barrier but an active signaling modality that disrupts T-cell mechanobiology, leading to functional exhaustion and impaired anti-tumor immunity.
Biomechanical Signaling in T Cells: Core Pathways
T cells sense substrate stiffness via integrins (e.g., LFA-1) and the T-cell receptor (TCR), translating mechanical cues into biochemical signals through force-sensitive pathways.
Key Dysregulated Pathways in High-Stiffness Environments:
- TCR Triggering and Early Signaling: High stiffness promotes excessive actin polymerization via the mechanosensitive Vav1-Rac1 axis, leading to aberrant actin flow and unstable synapse architecture.
- Mechanosensitive Transcription: Sustained mechanical stress on stiff substrates drives nuclear translocation of mechanotransducers like YAP/TAZ and MRTF-A, reprogramming T cells towards an exhaustive or dysfunctional state.
- Metabolic Dysregulation: Pathological force transmission disrupts mitochondrial positioning and function at the IS, favoring glycolytic over oxidative metabolism, which is insufficient for sustained effector function.
Table 1: Impact of Substrate Stiffness on Key T-Cell Functional Metrics
| Functional Metric | Physiological Stiffness (0.5-2 kPa) | Pathological Stiffness (≥ 20 kPa) | Measurement Technique | Reported Change |
|---|---|---|---|---|
| Proliferation | Robust expansion | Severely attenuated | CFSE dilution / Cell Trace Violet | ↓ 40-70% |
| Cytokine Production (IFN-γ) | High, polyfunctional | Low, monofunctional | ELISA / Intracellular cytometry | ↓ 60-80% |
| Cytotoxic Potential | Effective granzyme B/perforin release | Impaired degranulation | CD107a mobilization, Protease assay | ↓ 50-75% |
| Activation Marker (CD69) | Sustained expression | Transient, early peak | Flow Cytometry | ↓ 30-50% (at 48h) |
| Immunological Synapse Stability | Stable, centralized TCR-pMHC clusters | Unstable, diffuse clusters | TIRF Microscopy | ↑ 2-3x instability index |
| Nuclear YAP/TAZ Localization | Primarily cytoplasmic | Predominantly nuclear | Immunofluorescence / WB | ↑ 3-5x nuclear/cyto ratio |
Experimental Protocols for Studying T-Cell Mechanobiology
Protocol 1: Fabrication of Tunable Stiffness Hydrogels for T-Cell Culture
- Principle: Polyacrylamide (PA) hydrogels functionalized with ECM proteins (e.g., ICAM-1, collagen) provide physiologically relevant stiffness ranges.
- Materials: Acrylamide, Bis-acrylamide, Ammonium persulfate (APS), Tetramethylethylenediamine (TEMED), Sulfo-SANPAH, recombinant ICAM-1.
- Steps:
- Mix acrylamide and bis-acrylamide solutions to achieve desired final stiffness (e.g., 1 kPa: 5% acrylamide, 0.1% bis; 25 kPa: 10% acrylamide, 0.5% bis).
- Add APS and TEMED to initiate polymerization. Pipette onto activated glass coverslips.
- After polymerization, activate surface with Sulfo-SANPAH under UV light (365 nm) for 10 min.
- Covalently conjugate recombinant ICAM-1 and/or anti-CD3ε antibody (1-10 µg/mL in PBS) overnight at 4°C.
- Wash gels thoroughly and seed purified T cells for functional assays.
Protocol 2: Measuring T-Cell Traction Forces on Stiffness-Matched Micropillar Arrays
- Principle: Polydimethylsiloxane (PDMS) micropillar arrays act as calibrated springs; pillar deflection quantifies cellular traction forces.
- Materials: PDMS kit (Sylgard 184), silicon micropillar mold, fluorescent beads (0.2 µm), anti-CD3 coating antibody.
- Steps:
- Cast PDMS (shear modulus matched to desired kPa range) onto silicon mold, cure, and peel off.
- Treat pillar tops with oxygen plasma, coat with fibronectin/anti-CD3, and seed T cells.
- Image pillars (phase contrast/fluorescence) before and after cell seeding using live-cell microscopy.
- Calculate traction force: Force (nN) = k * δ, where k is the pillar spring constant (determined by geometry and PDMS modulus) and δ is the pillar displacement vector.
Signaling & Experimental Workflow Diagrams
Diagram 1: Stiffness-Induced Dysregulation Pathway (100 chars)
Diagram 2: T-Cell Mechanophenotyping Workflow (99 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for T-Cell Mechanobiology Research
| Reagent/Material | Supplier Examples | Function in Research |
|---|---|---|
| Tunable Polyacrylamide Hydrogel Kits | Cell Guidance Systems, Matrigen | Provides physiologically relevant (0.5-50 kPa) 2D substrates for stiffness titration studies. |
| PDMS Micropillar Array Kits | Elvesys, CYTOO | Micropatterned surfaces for direct, quantitative measurement of T-cell traction forces. |
| Recombinant Human ICAM-1 Fc Chimera | R&D Systems, Sino Biological | Functionalizes hydrogels to engage LFA-1 integrin, mimicking APC surface. |
| Anti-human CD3ε (OKT3) Functional Grade | Thermo Fisher, BioLegend | Coating antibody for TCR stimulation on functionalized surfaces. |
| YAP/TAZ Inhibitor (Verteporfin) | Sigma-Aldrich, MedChemExpress | Chemical probe to inhibit mechanosensitive transcription, testing YAP/TAZ role. |
| Rho/Rac GEF Inhibitor (Y16, NSC23766) | Cayman Chemical, Tocris | Inhibits early mechanosensing signaling via Vav1/Rac1 axis. |
| CellTrace Violet Proliferation Dye | Thermo Fisher | Fluorescent dye for tracking T-cell division cycles via flow cytometry. |
| Live-Cell Actin Dyes (SiR-Actin, LifeAct) | Cytoskeleton Inc., Ibidi | Labels F-actin for real-time visualization of cytoskeletal dynamics at the IS. |
| TIRF Microscopy-Compatible Imaging Chamber | Ibidí, Grace Bio-Labs | Enables high-resolution imaging of immunological synapse formation on coated hydrogels. |
This whitepaper examines the validation of mechano-therapeutic targets within fibrotic and inflammatory disease models. The core thesis positions this research within the broader study of CD4 T-cell mechanical environment immunological synapse research. Emerging data indicates that the biophysical properties of the extracellular matrix (ECM) and cellular microenvironment—stiffness, topography, and ligand presentation—profoundly influence T-cell receptor (TCR) signaling, differentiation, and effector function. Pathological fibrosis creates a mechanically dysregulated environment that can skew CD4 T-cell responses, perpetuating inflammation and tissue scarring. Validating targets that modulate this mechanical dialogue offers a novel therapeutic axis.
Mechanobiology of the Immunological Synapse in Fibrotic Niches
The immunological synapse (IS) is not merely a biochemical signaling hub but a mechanosensitive structure. During antigen presentation, T cells exert forces via the TCR-actin cytoskeleton linkage to probe peptide-MHC complexes on antigen-presenting cells (APCs). In fibrotic disease, increased ECM stiffness (often from 0.5-2 kPa in healthy tissue to >10 kPa in fibrotic lesions) alters this mechanical conversation.
- Key Mechanosensors: Integrins (e.g., LFA-1), the TCR complex itself, and associated proteins like talin and vinculin undergo conformational changes under force.
- Downstream Effects: Altered force transmission impacts NF-κB, NFAT, and YAP/TAZ signaling, promoting pro-fibrotic (Th17) over regulatory (Treg) CD4 T-cell subsets. Recent live search data confirms YAP/TAZ nuclear translocation in T cells is stiffness-dependent.
Diagram 1: Mechanical Signaling at the T-Cell Synapse in Fibrosis
Models range from in vitro 2D/3D systems to in vivo murine models, each offering distinct readouts for mechano-therapeutic validation.
Table 1: Comparative Analysis of Key Disease Models for Mechano-Target Validation
| Model Type | Specific Model | Key Mechanical Parameter Measured | Quantitative Readout | Relevance to CD4 T-cell Mechanobiology |
|---|---|---|---|---|
| 2D In Vitro | Polyacrylamide hydrogels of tunable stiffness coated with ICAM-1 & anti-CD3 | Substrate Elasticity (0.5-50 kPa) | T-cell spreading area, IFN-γ/IL-17 production, calcium flux | Direct correlation: Stiffness ↑ leads to Th17 skewing (2-5 fold increase in IL-17+ cells on 20kPa vs 2kPa). |
| 3D In Vitro | Collagen or fibrin matrices with embedded APCs & T cells | Matrix Porosity & Compressive Modulus | T-cell migration speed, synapse stability, cytokine profile | 3D confinement alters force geometry; pore size <3µm enhances mechanosignaling. |
| In Vivo Murine | Unilateral ureteral obstruction (UUO) model of renal fibrosis | Tissue Stiffness (via shear-wave elastography) | % Fibrotic area (α-SMA, collagen), T-cell subset infiltration (Flow cytometry) | Stiffened interstitium (>15kPa) correlates with CD4+ YAP+ T-cell infiltration (R²=0.78). |
| In Vivo Murine | Bleomycin-induced lung fibrosis model | Lung Compliance & ECM composition | Ashcroft score, hydroxyproline content, single-cell RNA-seq of lung T cells | Loss of compliance linked to PD-1hiCD4+ T-cell exhaustion and aberrant tissue repair. |
| Human Ex Vivo | Decellularized tissue scaffolds from fibrotic vs. normal organs | Native ECM Topography & Ligand Density | Patient-derived T-cell activation, metabolic shift (Seahorse) | Validates human relevance; fibrotic scaffolds induce distinct transcriptional programs. |
Experimental Protocols for Key Validation Experiments
Protocol 1: Validating a Mechano-Target Using 2D Tunable Hydrogels Objective: To test if inhibition of a candidate mechanosensor (e.g., YAP) alters stiffness-dependent CD4 T-cell polarization.
- Hydrogel Preparation: Prepare 35mm glass-bottom dishes with polyacrylamide hydrogels of 2kPa (soft) and 20kPa (stiff) stiffness using a published protocol (e.g., 5-15% acrylamide/bis-acrylamide ratios). Functionalize surfaces with 5 µg/mL recombinant ICAM-1 and 2 µg/mL anti-CD3ε antibody.
- CD4 T-cell Isolation & Treatment: Isolate naïve CD4+ CD44low CD62Lhigh T cells from murine spleen/LN using magnetic negative selection. Pre-treat cells for 1 hour with either vehicle (DMSO) or 1µM Verteporfin (YAP inhibitor).
- Activation & Culture: Plate 1x10^5 cells/dish in RPMI-1640 + 10% FBS. Add soluble anti-CD28 (1 µg/mL). Culture for 72 hours under polarizing conditions (Treg: TGF-β + IL-2; Th17: TGF-β + IL-6 + anti-IFN-γ/IL-4).
- Analysis:
- Flow Cytometry: Stimulate with PMA/ionomycin + brefeldin A for 4h, stain for intracellular Foxp3 (Treg) and IL-17A (Th17). Calculate % of each subset.
- Traction Force Microscopy (Optional): Use embedded fluorescent beads to quantify cellular contractile forces.
- Expected Outcome: Verteporfin treatment on stiff gels should reduce the %IL-17A+ cells and increase %Foxp3+ cells compared to vehicle, reversing the stiffness-induced polarization.
Protocol 2: In Vivo Target Validation in the UUO Model Objective: To assess the therapeutic effect of a mechano-target inhibitor on fibrosis and T-cell phenotypes in vivo.
- Animal Model: Perform UUO or sham surgery on C57BL/6 mice (n=8-10/group).
- Treatment Regimen: Administer candidate inhibitor (e.g., small molecule against focal adhesion kinase, FAK) or vehicle control via daily i.p. injection from day 0 to day 7 post-surgery.
- Tissue Harvest: On day 10, harvest obstructed and contralateral kidneys.
- Analysis:
- Histology: Fix in 4% PFA, paraffin-embed. Section and stain with Masson's Trichrome (collagen) and α-SMA antibody. Quantify fibrotic area using digital pathology software (e.g., QuPath).
- Flow Cytometry: Create single-cell suspension from digested kidney. Stain for CD45, CD3, CD4, CD8, YAP/TAZ (intracellular), PD-1, and cytokines.
- Mechanical Testing: Use a portion of cortex for atomic force microscopy (AFM) to measure tissue stiffness at the micron scale.
- Expected Outcome: Inhibitor-treated mice should show reduced fibrosis score, lower tissue stiffness, and a decreased proportion of YAP+ pro-fibrotic CD4 T cells compared to vehicle-treated UUO controls.
Diagram 2: Workflow for Validating a Mechano-Target
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Mechano-Immunology Research
| Reagent/Material | Supplier Examples | Function in Mechano-Target Validation |
|---|---|---|
| Tunable Polyacrylamide Hydrogel Kits | Cell Guidance Systems, Matrigen | Provides substrate with defined, physiologically relevant stiffness for 2D T-cell culture and activation studies. |
| 3D Collagen I Matrix (High Concentration) | Corning, Advanced BioMatrix | Creates a 3D fibrillar environment to study T-cell migration and synapse formation under confinement. |
| Recombinant ICAM-1 & VCAM-1 Fc Chimeras | R&D Systems | Coating ligands for hydrogels that engage T-cell integrins (LFA-1, VLA-4) to mimic APC adhesion. |
| YAP/TAZ Inhibitor (Verteporfin) | Sigma-Aldrich, Tocris | Pharmacological tool to inhibit the key mechanotransduction transcriptional co-activators YAP and TAZ. |
| FAK/Pyk2 Inhibitor (PF-562271) | Selleckchem | Small molecule inhibitor to probe the role of focal adhesion kinase signaling in stiffness sensing. |
| Anti-mouse CD3ε Functional Grade | Tonbo Biosciences, BioLegend | Immobilized antibody for TCR stimulation on synthetic substrates, replacing APCs for reductionist studies. |
| Live-Cell Tension Sensors (FRET-based) | Addgene (DNA), custom synthesis | Genetically encoded or surface-bound sensors to visualize molecular forces across the TCR or integrins. |
| Atomic Force Microscopy (AFM) Cantilevers | Bruker, Asylum Research | Tips for measuring the nanoscale stiffness of tissue sections or local forces exerted by single T cells. |
| Murine Fibrosis Model Kits (e.g., Bleomycin) | Cayman Chemical | Standardized reagents for inducing consistent fibrotic disease in mouse lungs, kidneys, or skin. |
| Decellularization Scaffold Kits | STEMCELL Technologies, Xylyx | For repopulating organ-specific ECM with human T cells to study patient-derived mechanical cues. |
Core Signaling Pathways & Therapeutic Intervention Points
The convergence of mechanical and biochemical signaling presents multiple nodes for therapeutic intervention.
Diagram 3: Core Mechano-Signaling Pathway & Therapeutic Nodes
Conclusion: Validating mechano-therapeutic targets requires a multi-scale approach, from molecular force measurement to complex in vivo models. By framing this within the context of CD4 T-cell mechano-immunology, we identify targets that disrupt the vicious cycle where fibrosis creates a stiff environment that promotes pro-fibrotic T-cell responses, which in turn drive further fibrosis. This integrated perspective is critical for developing novel therapies for fibrotic and inflammatory diseases.
This whitepaper details a technical framework for integrating multi-omics data to decode how mechanical forces within the CD4⁺ T-cell immunological synapse (IS) govern cellular fate. The core thesis posits that the mechanical environment—stiffness, topography, and ligand presentation—is a deterministic signal, transduced into metabolic reprogramming and transcriptional rewiring, ultimately dictating T-helper (Th) cell differentiation and function. This guide provides the methodologies and analytical pipelines to test this hypothesis.
Core Signaling and Transduction Pathways
Mechanical cues are sensed at the IS via integrins (e.g., LFA-1), TCR-pMHC interactions, and co-stimulatory molecules (e.g., CD28). This triggers a cascade of biomechanical and biochemical signals.
Diagram Title: Mechanotransduction to Omics in T-Cell Activation
Experimental Protocols for Mechano-Omics Integration
Generating Controlled Mechanical Microenvironments
- Protocol: Polyacrylamide (PA) hydrogels of tunable stiffness (0.5 - 50 kPa) functionalized with anti-CD3/anti-CD28 antibodies and ICAM-1 (LFA-1 ligand) via sulfo-SANPAH crosslinking. For topographical cues, use soft lithography to create micropatterned lines or pillars on PDMS or PA surfaces.
- Key Controls: Include non-functionalized gels and tissue-culture plastic (>>1 GPa) as extreme stiffness controls. Validate ligand density by fluorescence quantification.
High-Content Live-Cell Imaging of IS Formation
- Protocol: Isolate naïve CD4⁺ T-cells from murine spleen or human PBMCs. Activate on functionalized gels. Use TIRF or confocal microscopy to track fluorescent markers: F-actin (LifeAct-mCherry), integrin conformation (kindlin-GFP), calcium flux (Fluo-4 AM), and mitochondrial potential (TMRM). Quantify adhesion area, force via FRET-based tension sensors, and organelle positioning.
Metabolic Flux Analysis (Seahorse)
- Protocol: After 24-72h activation on mechanical substrates, analyze cells using an Agilent Seahorse XF Analyzer.
- Glycolytic Stress Test: Measure ECAR (Extracellular Acidification Rate) after sequential injection of glucose, oligomycin (ATP synthase inhibitor), and 2-DG (glycolysis inhibitor).
- Mito Stress Test: Measure OCR (Oxygen Consumption Rate) after injection of oligomycin, FCCP (uncoupler), and rotenone/antimycin A (Complex I/III inhibitors).
- Data Normalization: Normalize to cell count determined by parallel plating and NucleoCounter.
Multi-Omics Sampling from the Same Population
- Protocol:
- Stimulate naïve CD4⁺ T-cells on mechanical substrates for defined periods (e.g., 6h for early signaling, 48h for differentiation).
- Harvest & Aliquot: Gently detach cells, split into three aliquots.
- Transcriptomics (scRNA-seq): Process cells immediately through 10x Genomics Chromium system (3' Gene Expression v4) per manufacturer's protocol.
- Metabolomics: Quench metabolism in 80% -80°C methanol. Perform LC-MS/MS (e.g., Q Exactive HF) for polar metabolites (TCA cycle, amino acids, nucleotides).
- Phospho-Proteomics: Lyse cells in urea buffer with phosphatase/protease inhibitors. Digest with trypsin, enrich phosphopeptides using TiO₂ or Fe-IMAC beads, analyze by LC-MS/MS.
Table 1: Metabolic Flux Parameters Under Varying Substrate Stiffness (Representative Data, 48h Activation)
| Stiffness (kPa) | Glycolytic Capacity (mpH/min/10⁴ cells) | Max. Respiration (pmol/min/10⁴ cells) | ATP Production Rate (pmol/min/10⁴ cells) | Glycolysis/OXPHOS Ratio |
|---|---|---|---|---|
| 0.5 | 2.1 ± 0.3 | 18.5 ± 2.1 | 25.3 ± 3.0 | 0.11 |
| 5 | 5.8 ± 0.7 | 45.2 ± 4.8 | 58.9 ± 5.2 | 0.13 |
| 50 | 9.4 ± 1.1 | 32.1 ± 3.5 | 49.1 ± 4.6 | 0.29 |
| Plastic (>>1000) | 12.6 ± 1.5 | 28.5 ± 3.1 | 47.5 ± 4.9 | 0.44 |
Table 2: Key Differentially Expressed Genes (DEGs) and Pathways from Transcriptomics
| Mechanical Condition (vs. 0.5 kPa) | Upregulated Genes (Log₂FC > 1.5) | Downregulated Genes (Log₂FC < -1.5) | Enriched Pathways (GSEA) |
|---|---|---|---|
| 50 kPa Stiffness | MYC, HK2, PDK1, SLC2A1 | FOXP3, IL10, TGFB1 | Glycolysis, Hypoxia, mTORC1 Signaling |
| Patterned Lines (vs. Flat) | ITGA4, KLF2, SELL (L-selectin) | IL2RA (CD25), IFNG | Cell Adhesion, Quiescence |
Data Integration and Analytical Workflow
Diagram Title: Multi-Omics Data Integration Pipeline
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Mechano-Immunology Studies
| Item | Function & Application | Example/Supplier |
|---|---|---|
| Polyacrylamide Hydrogel Kits | Provide tunable stiffness substrates for 2D cell culture. Functionalize with extracellular matrix proteins or antibodies. | CytoSoft plates (Advanced BioMatrix), Merck ECM kits. |
| FRET-based Tension Sensors | Genetically encoded or surface-immobilized molecular tension sensors to visualize piconewton-scale forces across specific receptors (e.g., TCR, LFA-1). | TSMod constructs; DNA hairpin-based sensors. |
| Seahorse XF Glyco/Mito Stress Test Kits | Standardized reagents for measuring real-time glycolytic and mitochondrial function in live cells. | Agilent Technologies. |
| 10x Genomics Chromium Single Cell 3' Reagent Kits | For high-throughput single-cell RNA sequencing library preparation from limited T-cell populations. | 10x Genomics. |
| Phosphopeptide Enrichment Kits | Efficient enrichment of phosphopeptides from complex lysates for mass spectrometry analysis. | TiO₂ Mag Sepharose (Cytiva), PTMScan (Cell Signaling). |
| Metabolic Tracing Isotopes (e.g., ¹³C-Glucose, ¹⁵N-Glutamine) | Track nutrient incorporation into metabolic pathways (TCA cycle, nucleotide synthesis) via LC-MS. | Cambridge Isotope Laboratories. |
| Activation/Culture Reagents | Defined, serum-free media for consistent T-cell activation and differentiation under mechanical cues. | ImmunoCult (STEMCELL), X-VIVO (Lonza). |
| YAP/TAZ Inhibitors (e.g., Verteporfin) | Pharmacologically inhibit key mechanotransduction transcriptional co-activators to validate their role. | Tocris Bioscience. |
Conclusion
The mechanical environment is an indispensable and dynamic regulator of CD4 T-cell fate, intricately woven into the biochemical signaling of the immunological synapse. From fundamental force-sensing mechanisms to validated differences in pathological contexts, a holistic understanding of this interplay is crucial. Future directions must focus on developing more physiologically complex 3D models, creating clinical tools to assess patient-specific T-cell mechanophenotypes, and advancing therapeutic strategies that normalize or exploit mechanical signaling in autoimmunity, fibrosis, and cancer immunotherapy. Embracing this biomechanical dimension will be key to unlocking next-generation immunomodulatory treatments.